
Concept explainers
Interpretation:
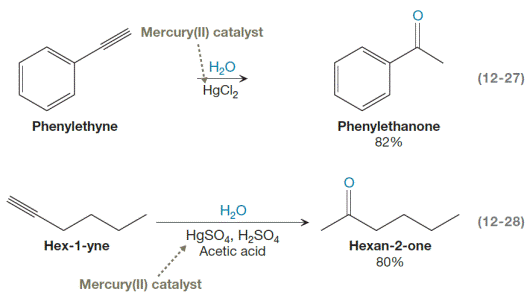
Equations 12-27 and 12-28 show the overall products of hydration of the
Concept introduction:
Alkynes can undergo Markovnikov addition of water via oxymercuration. Hydration of an alkyne leads to an initial enol, which tautomerizes into the more stable keto form. A
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Instructions: Show the major products of these reactions. Show the COMPLETE reaction.arrow_forwardComplete the following reaction schemes. Some may take more than one step and some may have several routes to the product.arrow_forwardI understand what reagents and or solvents can be used to get from an alkene to an alkyne but if someone could please show me step by step how each is used to perform the actual synthesis? Like how does each thing work to get to the next portion of the synthesis. I am truly struggling. thank youarrow_forward
- Show the alpha hydrogens that are on each of the following alkenes. (please also say the number of alpha hydrogens that are on each of the following alkenes). Then, rank the alkenes in order of their heats of hydrogenation (1=lowest, 4=highest).arrow_forwardShow the major products of these reactoions, and please show the complete reactionarrow_forwardShow two different ways to make an epoxide from a trans alkene, one that gives the trans epoxide and one that give the cis epoxidearrow_forward
- There are four different starting molecules that one might use to synthesize the illustrated alkyl halide as the major product using an electrophilic addition reaction. Please draw all four of them.arrow_forwardComplete synthesis in 2-3 stepsarrow_forwardshow the arrows of this reaction mechanism, the major product should show a cyclopentane with 2 methyl groups on carbon 2 and a double-bonded O on carbon 1arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
