
Interpretation:
The mechanism for the reaction in Equation 12-17 is as follows, but the curved arrows have been omitted. The mechanism by drawing in the curved arrows is to be completed. Also, the name of each elementary step below the appropriate reaction arrow is to be written.
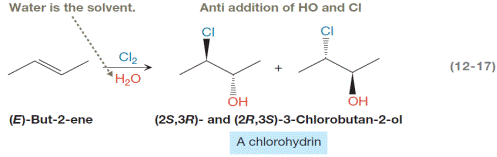
Concept introduction:
A chlorohydrin is produced when an
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Show every step of the mechanism for the following reactionarrow_forwardPlease show the mechanism and product for this reactionarrow_forwardHelp please. Draw the curved arrow notation and products for the each elementary step described by the sequence shown here.Note that the products of each step should be used as the reactants in the next step, and you may need to draw in additional reagents as directed. Remember to click on each box to see step-specific instructions. Draw H3CH2C–, and then add the curved arrow notation showing a nucleophilic addition.arrow_forward
- Submit the mechanism for the following reaction. Use the mechanism you draw to match this reaction with the correct energy diagram.arrow_forwardDraw the product, state the reaction type and draw out the key steps of the mechanism.arrow_forwardFor the following reaction, draw a complete reaction mechanism using curvy arrows being sure to show all important intermediates and the final product.arrow_forward
- The reaction shown here is a halosulfonation, which is a useful variation of the sulfonation reaction. Draw the complete mechanism for this reaction.arrow_forwardSHOW THE MECHANISM AND THE MAJOR PRODUCT/Sarrow_forwardDraw the complete mechanism of each pair of reactants including any favorable rearrangements and all important resonance structures of all intermediates. a. Which reaction has a lower PE carbocation intermediate? b. Draw an energy diagram showing the reaction profiles of both reactions in the previous question. Use a dotted line for the first pair of reactants and a solid line for the second pair of reactants. (Assume the energy of the starting materials and products are the same for both pairs and the reactions are neither uphill nor downhill on net. c. Mark points on the energy diagram corresponding to each carbocation in your mechanisms.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
