
Concept explainers
Interpretation:
Solid, liquid, gas, triple point and critical point must be labeled in the given phase diagram.
Concept introduction:
Phase diagram is a graphical representation of phase change with pressure and temperature.
Triple point is the condition at which solid, liquid and gas coexist.
Critical point is the temperature at which liquid is in equilibrium with vapor.
Answer to Problem 81A
Solid, liquid, gas, triple point and critical points are labeled on the phase diagram.
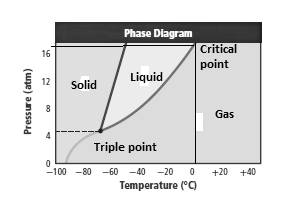
Explanation of Solution
As can be seen from the phase diagram solid, liquid and gas phase are separately shown. Along X-axis and Y-axis temperature and pressure are plotted. Triple point is the black dot where all the solid, liquid and gas lines intersect each other. This point represents that all the phase are present at this point. Critical temperature is shown as the boundary of liquid and gas as at critical point liquid is in equilibrium with gas.
Phase diagram is a graphical representation of phase change of a substance along with pressure and temperature.
Chapter 12 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
CHEMISTRY-TEXT
Chemistry: A Molecular Approach (4th Edition)
Organic Chemistry (9th Edition)
Organic Chemistry (8th Edition)
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





