
Concept explainers
a.
For the below compound (Enantiomer) Fischer projection has to be drawn.
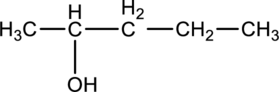
Concept introduction:
Fischer projections:
In Fischer projection, the chirality center of the organic compounds are drawn using the following convention. First, the carbon atom is placed at the meeting point of the two lines of the cross. In a tetrahedron structure, the horizontal bonds indicate forward and denoted by wedges. The vertical bonds indicate backward and denoted by dashed lines. Finally, Fischer projection structure is abbreviated by cross formula.
Enantiomers:
Enantiomers are mirror image of the other and these are chiral molecules,but that are non superimposableon one another.
b.
Interpretation:
For the below compound (Enantiomer) Fischer projection has to be drawn.
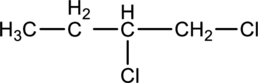
Concept introduction:
Refer to part ‘a.’.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Locate the stereogenic centers in each compound.arrow_forwardFor those that have only one stereocenter draw Fischer projection(s) of the S-stereoisomer placing the 4 and 2 positions on the vertical line.arrow_forwardPart B. Mark each chirality center in each molecule with an asterisk (*).arrow_forward
- Captopril is a drug used to treat high blood pressure and congestiveheart failure.Draw the enantiomer of captoprilarrow_forwardDraw the enantiomer of each compound.arrow_forwardRe-draw each Fischer projection formula using wedges and dashed wedgesfor the stereogenic center, and label the center as R or S.arrow_forward
- Part B. Mark each chirality center in each molecule with an asterisk (*). Illustrate onto the image Cannot be hand-drawnarrow_forwardLabel the two stereogenic centers in each compound and draw all possible stereoisomers.arrow_forwardConvert each compound to a Fischer projection, and label each stereogenic center as R or S.arrow_forward
