
Concept explainers
a.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined
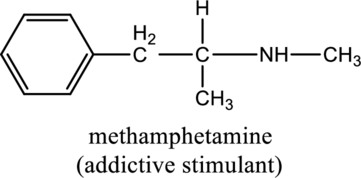
Concept introduction:
Chirality center:
When a carbon atom is bonded to four different groups, then that carbon is named as chiral carbon and it is called as chirality center. Generally, the carbon atom contains tetrahedral valency, therefore it bonded to four atoms or molecules. The chiral center is bonded with four different elements or groups. If the single carbon atom is bonded to two or more identical group or atom, then it is also not considered as chiral carbon. In addition to this, if the carbon atom contains multiple bond then it does not able to bond with four different groups then it is not considered to be as a chirality center.
b.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined
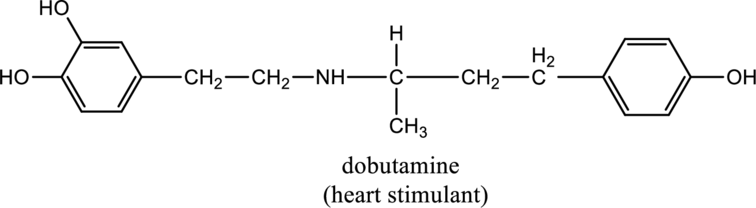
Concept introduction:
Refer to part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- One of the analgesics has a chiral center. Which compound is it? One of the two enantiomers is far more effective at reducing pain than the other.arrow_forwardFluocinolone how many chiral centers? compound numbering Fluocinolone.?arrow_forwardEphedrine (2‑methylamino‑1‑phenylpropanol) is a drug that is used as a decongestant and bronchodilator to treat, among other conditions, the congestion due to the common cold. Place an asterisk (*) next to the chirality center(s) in the ephedrine molecule.arrow_forward
- Translate the ball-and-stick model to a shorthand structure, and label the two chirality centers in vitamin K. Vitamin K is essential to blood clot formation. A defi ciency of this vitamin may lead to excessive and often fatal bleeding.arrow_forwardConvert each compound to a Fischer projection formula.arrow_forwardDetermine the relationship between the two compounds: A. Same compound B. Enantiomers C. Diastereomers D. Constitutional isomersarrow_forward
- Optically active one compound. Optically active pair of diastereomers. Optically inactive one compound. Optically inactive pair of diastereomers. Optically active pair of enantiomers. Optically inactive pair of enantiomers.arrow_forwardConvert each ball-and-stick model to a Fischer projection.arrow_forward(a) assign R or S configuration to each chiral center, (b) Which compound are enantiomers? (c) Which compounds are diastereomers?arrow_forward
- A(n) ________ is an achiral compound that contains chiral centers but is superimposable on its mirror image. A) constitutional isomers B) conformational isomers C) enantiomers D) diastereomers E) meso compoundsarrow_forward(i) What is meant by chirality of a compound? Give an example.(ii) Which one of the following compounds is more easily hydrolyzed by KOH and why?CH3CHClCH2CH3 or CH3CH2CH2Clarrow_forwardDetermine the relationship between the two drawings: A. Enantiomers B. Same compound C. Constitutional isomers D. Diastereomersarrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


