
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers and the type of alcohol should be determined.
Concept introduction:
Chirality center:
When a carbon atom is bonded to four different groups, then that carbon is named as chiral carbon and it is called as chirality center. Generally, the carbon atom contains tetrahedral valency, therefore it bonded to four atoms or molecules. The chiral center is bonded with four different elements or groups. If the single carbon atom is bonded to two or more identical group or atom, then it is also not considered as chiral carbon. In addition to this, if the carbon atom contains multiple bond then it does not able to bond with four different groups then it is not considered to be as a chirality center.
Classification of alcohol:
Generally the alcohol group is bonded with minimum one alkyl group. The alcohol group is classified into three types, such as primary
Explanation of Solution
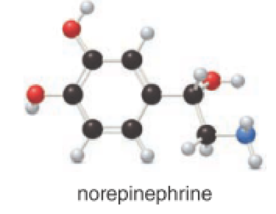
Given compound is norepinephrine,
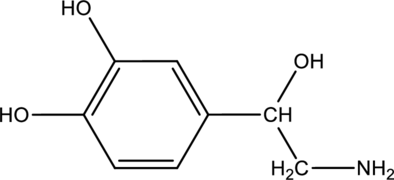
In the given drug (i,e) norepinephrine, the
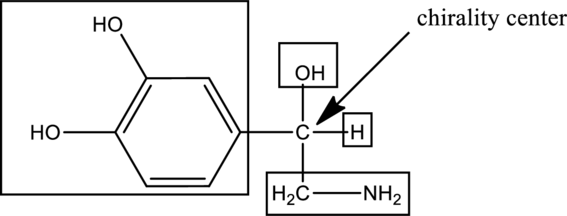
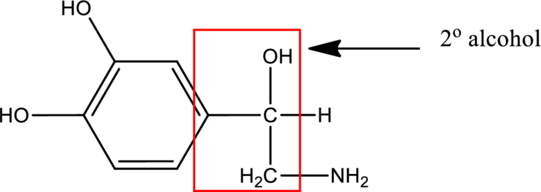
In the below compound,
Want to see more full solutions like this?
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Label each of the following structures as a hemiacetal, hemiketal, acetal, ketal, or none of these: a. c. b.arrow_forward18-19 The following compounds have approximately the same molecular weight: hexanoic acid, heptanal, and 1-heptanol. Arrange them in order of increasing boiling point.arrow_forward17-15 Draw structural formulas for the four aldehydes with the molecular formula C5H10O. Which of these aldehydes are chiral?arrow_forward
- What happens when phenol is oxidized by Na2Cr2O7/H2SO4?arrow_forwardIdentify the IUPAC name of the given compound. a) 2-methyl-3-amino-1-cyclopentanone b) 1-amino-2-methyl-3-cyclopentanone c) 3-amino-2-methyl-1-cyclopentanone d) 2-amino-1-methyl-2-cyclopentanonearrow_forward(a) Identify the functional groups in the ball-and-stick model of pantothenic acid, vitamin B5. (b) At which sites can pantothenic acid hydrogen bond to water? (c) Predict the water solubility of pantothenic acid.arrow_forward
- What is the IUPAC name for CH3CH2CH2CH2OHCH3CH2CH2CH2OH? 5-pentanal pentanal 3-butanol 1-butanol 5-pentanol 1-pentanolarrow_forwardEphedrine (2‑methylamino‑1‑phenylpropanol) is a drug that is used as a decongestant and bronchodilator to treat, among other conditions, the congestion due to the common cold. Place an asterisk (*) next to the chirality center(s) in the ephedrine molecule.arrow_forward16-46 Arrange these three compounds in order of decreas ing ability to form intermolecular hydrogen bonds: CH3OH, CH3SH, and (CH3)2NH.arrow_forward
- Place an asterisk (*) next to the chirality center (sometimes called chiral atom) or centers in each compound. It is possible that a compound may not have a chiral center.arrow_forwardDraw the products formed when each alcohol is dehydrated with H 2SO 4. Use the Zaitsev rule to predict the major product when a mixture forms.arrow_forward3.A carbon atom that is bonded to four different groups is a/an A.achiral center B.chirality center C.symmetrical center D.symmetrical centerarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



