
Concept explainers
a.
Interpretation:
For the below ball and stick model, name has to be determined.
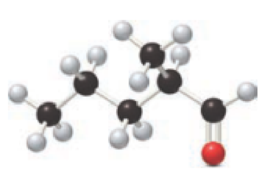
Concept Introduction:
Nomenclature of
Firstly, the functional group from the below structure will be identified as aldehyde and –al is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the -CHO group. In case of the cyclic compounds, numbering starts with –CHO group attached carbon, which is considered a primary carbon. Prefix changes if any other substituents are present in the structure other than –CHO group. In case of more than one substituents naming will be given in alphabetical order. For the parent
b.
Interpretation:
For the below ball and stick model, name has to be determined.
.
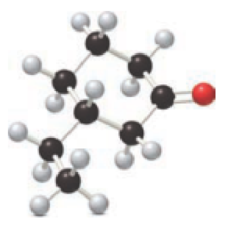
Concept introduction:
Naming of
Firstly, the functional group from the below structure will be identified as ketone and –one is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the ketone group. In case of the cyclic compounds, numbering starts with ketone group attached carbon, which is considered a primary carbon. Prefix changes if any other substituents are present in the structure other than ketone group. In case of more than one substituents naming will be given in alphabetical order.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Rank the labeled nitrogen atoms in each compound in order of increasing basicity. Histamine causes the runny nose and watery eyes associated with allergies, and trazodone is a drug used as a sedative and antidepressant.arrow_forwardGive the IUPAC name (including any E,Z designation) for each unsaturated aldehyde. Neral is obtained from lemon grass, and cucumber aldehyde (Problem 1.30) contributes to the aroma of a fresh mango.arrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forward
- Draw the hemiacetal and acetal formed when the carbonyl compound is treated with two equivalents of the given alcohol in the presence of H2SO4.arrow_forwardShow how to prepare pentanoic acid from each component. (a) 1-Pentanol (b) Pentanal (c) 1-Pentene (d) 1-Butanol (e) 1-Bromopropane (f) 1-Hexenearrow_forwardIn the presence of 1 equivalent NaCN, indicate which carbonyl group is preferentially attacked. Explain why your group of choice is preferentially attacked.arrow_forward
- Draw the products formed when each alcohol undergoes dehydration with TsOH, and label the major product when a mixture results.arrow_forwardWhat Grignard reagent and carbonyl compound are needed to prepare each alcohol?arrow_forwardwhich reaction will lead to the formation of sodium benzoate? a) phenol + NaOH b) benzoic acid + NaOH c) ethyl benzoate + NaOH d) both b and carrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


