
Concept explainers
a.
Interpretation:
From a given pair of compound, which one is higher boiling point has to be determined.
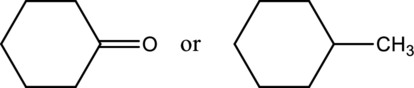
Concept introduction:
Boiling point of aldehyde, ketone, alcohol and hydrocarbons:
Carbonyl compounds such as
b.
Interpretation:
From a given pair of compound, which one is higher boiling point has to be determined.
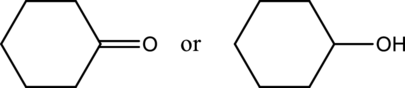
Concept introduction:
Refer to part a.
c.
Interpretation:
From a given pair of compound, which one is higher boiling point has to be determined.
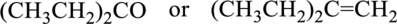
Concept introduction:
Refer to part a.
d.
Interpretation:
From a given pair of compound, which one is higher boiling point has to be determined.
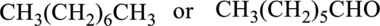
Concept introduction:
Refer to part a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Which group in each pair is assigned the higher priority? a. – CH3, – CH2CH3 b. – I, – Br c. – H, – D d. – CH2Br, – CH2CH2Br e. – CH2CH2Cl, – CH2CH(CH3)2 f. – CH2OH, – CHOarrow_forwardExplain why CH3CH2NHCH3 has a higher boiling point than (CH3)3N, even though they have the same molecular weight.arrow_forwardn-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forward
- Which member of each pair is more soluble in water? Why?(a) CH₃CH₂OCH₂CH₃(l) or CH₃CH₂OCH₃(g)(b) CH₂Cl₂(l) or CCl₄(l)arrow_forwardAnswer each question using the ball-and-stick model of compound A. Draw a constitutional isomer that contains an ether and give its IUPACname. Draw the products formed (including stereochemistry) when A istreated with each reagent: [1] NaH; [2] H2SO4; [3] POCl3, pyridine; [4]HCl; [5] SOCl2, pyridine; [6] TsCl, pyridinearrow_forwardDraw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each Wittig reagent.arrow_forward
- Using the cyclohexane with the C’s numbered as shown, draw a chair form that ts each description.a. The ring has an axial CH3 group at C1 and an equatorial OH on C2.b. The ring has an equatorial CH3 group on C6 and an axial OH group on C4.c. The ring has equatorial OH groups on C1, C2, and C5.arrow_forwardWhich group has the highest priority according to the Cahn, Ingold, Prelog rules? Select one: a. -CHO b. -OCOH c. -OCH3 d. - OCH2CH3 e. -COOCH3arrow_forwardDraw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forward
- Which group in following pair is assigned the higher priority? −CH2CH2Cl, −CH2CH(CH3)2arrow_forwardIsomers with the same boiling point and molecular weight but have different boiling points how is this possible ?arrow_forwardWhich compound will give a positive test with Br2/CH3Cl, a positive test with HCl/ZnCl2, and a positive test with CrO3/H2SO4?arrow_forward
