
Concept explainers
Interpretation:
The proton decoupled
Concept introduction:
The
Answer to Problem 13.63AP
The proton decoupled
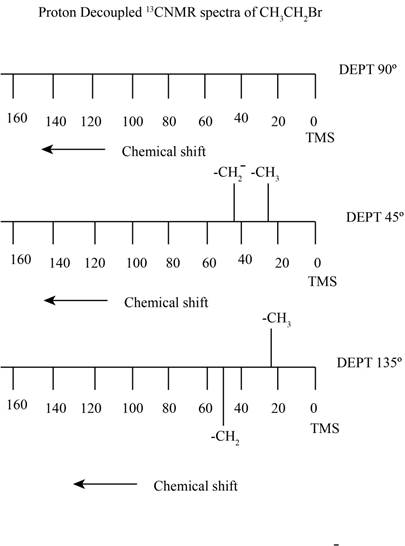
Explanation of Solution
The carbon atom exists in two isotopes
The relative intensity for the molecule in which no
The coefficient represents the number of atoms present of each element. Here the possibility of two
Here,
Here the possibility of two
The compound ethyl bromide contains two types of proton containing carbon. It gives rise to a quartet signal of
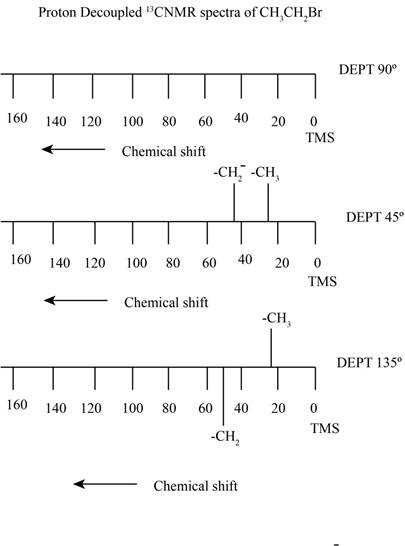
Figure 1
The proton decoupled
Want to see more full solutions like this?
Chapter 13 Solutions
ORGANIC CHEMISTRY (LL)+ SAPLING ACC >BI
- Explain all splitting patterns observed for absorptions between 0 and 8 ppm for the alcohol shown below. Which proton or protons will give rise to a doublet at 1.4 , which ones will give rise to a singlet at 2.7, which ones will give rise to a quartet at 4.7 and which ones will give rise to multiplets at 7.4?arrow_forward4. Why does a carbon to oxygen double bond absorption band have a greater intensity than a carbon to carbon double bond absorption band?arrow_forwardFill in the table by matching the numbers of the structure to their corresponding peaks of the C-13 spectra.arrow_forward
- What can be said about the ratios of the multiplet areas in the 13C spectra for trichloroethene; 1,2-dichloroethane; 1,1,1-trichloroethane; 1,1,2-trichloroethane.arrow_forwardHydrohalic acids (i.e., HCl, HBr, HI) add to the double bond of alkenes to yield alkyl halides. Shown below is the carbon NMR spectrum of an alkene of formula C5H10. What is the structure of this alkene?arrow_forwardWhich of the following compounds can have a broad absorbance band near 3300 cm-1 and 1650 cm-1?arrow_forward
- What characteristics of the methyl benzoate spectrum rule out an aldehyde or carboxylicacid functional group giving the absorption at 1723 cm-1?arrow_forwardPropose a structure consistent with each set of data. a.C9H10O2: IR absorption at 1718 cm−1b.C9H12: IR absorption at 2850–3150 cm−1arrow_forwardC8H8O produces an IR spectrum with 3063, 1686, 1646 cm signals. HNMR is a singlet at 2.6ppm (3H), and multiplet at 7.5 (5H). What is the productarrow_forward
- Can a low-resolution mass spectrometer distinguish between C2H5 + and CHO+ ?arrow_forwardThe infrared spectrum of CBr4 has a strong absorption at 667 cm-1. What is the correct assignment of this absorption?and why ? a.) A bending or bending mode. b.) The asymmetric stretching of the four C-Br bonds. c.) The symmetric stretching of the four C-Br bonds. d.) The stretching of a C-Br bond.arrow_forwardThe natural abundance of 13C is only 1.1%. Furthermore, its sensitivity in NMR spectroscopy (a measure of the energy difference between a spin aligned with or against an applied magnetic field) is only 1.6% that of 1H. What are the relative signal intensities expected for the 1H-NMR and 13C-NMR spectra of the same sample of Si(CH3)4?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



