
Concept explainers
(a)
Interpretation: The species which have same number of atoms and same number of valence electrons in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
(a)
Answer to Problem 65E
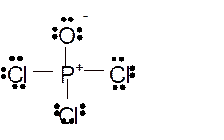
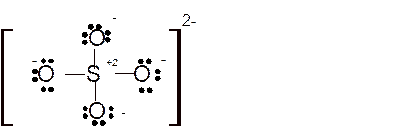
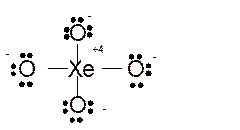
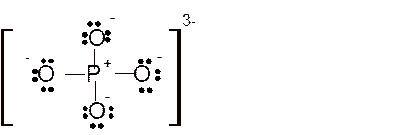
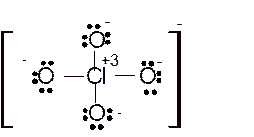
Hence here all
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
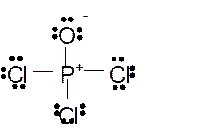
Total number of valence electrons in
Hence the best Lewis structure for
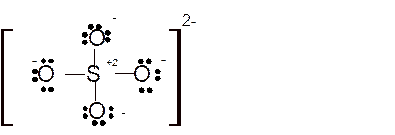
Total number of valence electrons in
Hence the best Lewis structure for
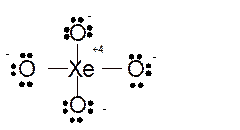
Total number of valence electrons in
Hence the best Lewis structure for
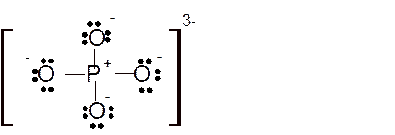
Total number of valence electrons in
Hence the best Lewis structure for
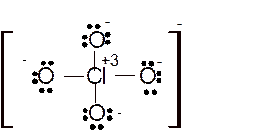
Hence here all
(b)
Interpretation: The species which have same number of atoms and same number of valence electrons in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
(b)
Answer to Problem 65E
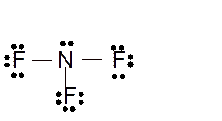
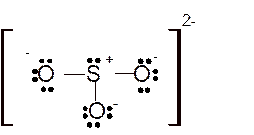
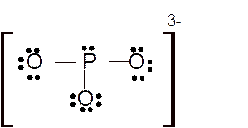
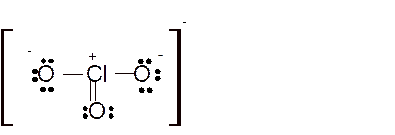
Hence here all
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
Total number of valence electrons in
Hence the best Lewis structure for
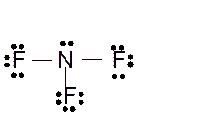
Total number of valence electrons in
Hence the best Lewis structure for
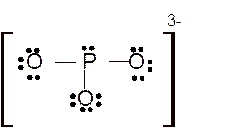
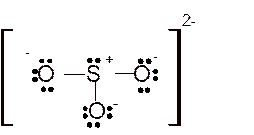
Total number of valence electrons in
Hence the best Lewis structure for
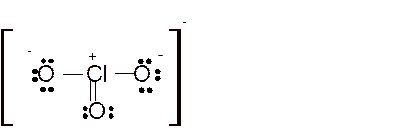
Hence here all
(c)
Interpretation: The species which have same number of atoms and same number of valence electrons in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
(c)
Answer to Problem 65E
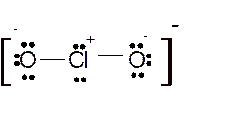
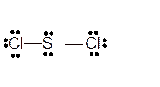
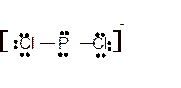
Hence here all
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
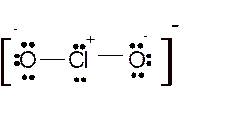
Total number of valence electrons in
Hence the best Lewis structure for
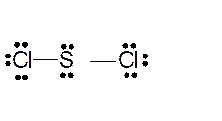
Total number of valence electrons in
Hence the best Lewis structure for
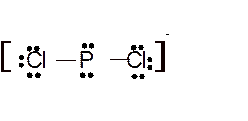
Hence here all
Want to see more full solutions like this?
Chapter 13 Solutions
Chemical Principles
- The Be2 molecule has been detected experimentally. It has a bond length of 2.45 Å and a bond dissociation energy of 9.46kJmol1 . Write the ground-state electron configuration of Be2 and predict its bond order using the theory developed in the text. Compare the experimental bonding data on Be2 with those recorded for B2,C2,N2, and O2 in Table 6.3. Is the prediction that stems from the simple theory significantly incorrect?arrow_forwardThe B2 molecule is paramagnetic; show how this indicates that the energy ordering of the orbitals in this molecule is given by Figure 6.18a rather than 6.18b.arrow_forwardCompare the electron density in the 1g and 1u* molecular orbitals for H2+ shown in Figure 6.5 with the classical model for bonding for H2+ summarized in Figure 3.22b. Which of these molecular orbitals describes the bond in H2+ ?arrow_forward
- Calcium carbide (CaC2) is an intermediate in the manufacturing of acetylene (C2H2) . It is the calcium salt of the carbide (also called acetylide) ion (C22) . What is the electron configuration of this molecular ion? What is its bond order?arrow_forwardIn a gaseous RbF molecule, the bond length is 2.2741010m . Using data from Appendix F and making the same oversimplified assumption as in the prior problem on the shape of the potential curve from Rb++F to an internuclear separation of 2.2741010m , calculate the energy in kJmol1 required to dissociate RbF to neutral atoms.arrow_forwardThe molecular orbital diagram of NO shown in Figure 10.47 also applies to OF. Draw the complete molecular orbital diagram for OF. What is the OF bond order? Figure 10.47 Molecular orbital diagram for nitric oxide (NO). The molecular orbital diagram for NO predicts a bond order of 2.5 and predicts that the molecule is paramagnetic with one unpaired electron. These predictions are verified by experimental measurements.arrow_forward
- • describe chemical bonding using a model based on the overlap of atomic orbitals and recognize sonic of the limitations of this simple model.arrow_forwardUse the data in Table 3.1 to plot the logarithm of ionization energy versus the number of electrons removed for Be. Describe the electronic structure of the Be atom.arrow_forwardCan a molecule with an odd number of electrons ever be diamagnetic? Explain why or why not.arrow_forward
- Explain why 1g is the ground state for H2+ . By combining your answer with the answer to Problem 5, what conclusions can you draw about the molecular orbital description of the bond in H2+ ?arrow_forward(a) Sketch the occupied MOs of the valence shell for the N2 molecule. Label the orbitals as or orbitals, and specify which are bonding and which are antibonding. (b) If one electron is removed from the highest occupied orbital of N2 , will the equilibrium NN distance become longer or shorter? Explain briefly.arrow_forwardExplain the difference between the and MOs for homonuclear diatomic molecules. How are bonding and antibonding orbitals different? Why are there two MOs and one MO? Why are the MOs degenerate?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





