
(a)
Interpretation:
The following molecule should be named:
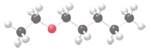
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
In ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the
(b)
Interpretation:
The following molecule should be named:
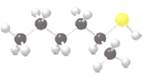
Concept Introduction:
Organic compounds which contains a sulfhydryl group (SH) bonded to a tetrahedral carbon is known as thiols. In the periodic table, sulfur is placed directly below to the oxygen, thiols can be considered as sulfur analogs of alcohols.
When naming the thiols, parent hydrocarbon is named as alkane and at the end -thiol suffix should be added. Main carbon chain is numbered by giving the SH group the lower number.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Classify each molecule as an aldehyde, ketone, or neither. AND Classify each molecule as an ester, ether, or neither.arrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 7: Put it all together and give the IUPAC name. You have determined the name of the longest carbon chain and the locations and name of the substituents. Complete the name of the molecule by adding in the hyphens and commas. 3ethyl223trimethyl5propyldecane 3-ethyl,2,2,3trimethyl-5propyldecane IUPAC name: Incorrectarrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 7: Put it all together and give the IUPAC name. You have determined the name of the longest carbon chain and the locations and name of the substituents. Complete the name of the molecule by adding in the hyphens and commas. 3ethyl223trimethyl5propyldecane JUPAC name:arrow_forward
- Encircle the functional groups, label each functional group with a letter, and identify the type or class of compounds represented by each functional group. 1. LSD, a hallucinogenic drug widely believed to be the inspiration behind the Beatles hit “Lucy in the Sky with Diamonds” 2. Methyljasmonate, a compound that belongs to a group called the pheromones. The male oriental fruit moth, Grapholitha molesta Busk., responds when a female moth emits this compound. 3.arrow_forwardH3C C CH₂CH3 CH₂MgBr+ Ether H₂Oarrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 6: Add the substituent locants or numbering. Now that you have identified the order of the substituents, you need to provide the locant, which indicates the number carbon on the main chain where each substituent is placed. Carbon 5 has only one substituent, so the name is represented by 5-propyl. Both carbons 2 and 3 have two substituents. What would be the correct naming for those carbons? The substituents should be named: 2,2-dimethyl and 3-methyl-3-ethyl 2,3-trimethyl and 3-ethyl 2,2,3-trimethyl and 3-ethyl 2-dimethyl and 3-methylethylarrow_forward
- What is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 3: Number the longest chain of carbon atoms so that the substituents have the lowest possible numbers. Carbon atoms that are not in the longest chain should not be numbered. Answer Bank 10 5 3 8 2.arrow_forwardWhat is the IUPAC name for the compound shown? Step I: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 4: Identify the substituents in the molecule. A. How many methyl substituents are in the compound? B. What prefix is needed for the methyl substituents? C. What are the remaining substituents? ethyl pentyl propyl butylarrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 5: Order the substituents. Since there are multiple substituents present, we must next consider the order the substituents in the name. The substituent order should be methyl, ethyl, followed by propyl ethyl, propyl, followed by trimethyl trimethyl, ethyl followed by propyl O ethyl, trimethyl, followed by propylarrow_forward
- Cyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?arrow_forwardOut of CH3—NH2 and (CH3)3N, which one has higher boiling point?arrow_forwardDraw an isomer of C5H10O that contains an alcoholarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





