
(a)
Interpretation:
The following compound should be labeled as water-soluble or water insoluble:


Concept introduction:
The solubility of a molecule depends on the polarity of the solvent and solution that means like dissolve like. In other words, the polar molecule is soluble in polar solvent only and in non-polar solvents, the soluble molecule will be non-polar in nature.
(b)
Interpretation:
The following compound should be labeled as water-soluble or water insoluble:
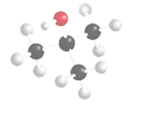

Concept introduction:
The solubility of a molecule depends on the polarity of the solvent and solution that means like dissolve like. In other words, the polar molecule is soluble in polar solvent only and in non-polar solvents, the soluble molecule will be non-polar in nature.
(c)
Interpretation:
The following compound should be labeled as water-soluble or water insoluble:
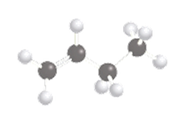

Concept introduction:
The solubility of a molecule depends on the polarity of the solvent and solution that means like dissolve like. In other words, the polar molecule is soluble in polar solvent only and in non-polar solvents, the soluble molecule will be non-polar in nature.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Classify each of the following solutions as saturated, unsaturated, or supersaturated based on the following observations made after adding a small piece of solid solute to the solution. a. The added solute rapidly dissolves. b. The added solute falls to the bottom of the container where it remains without any decrease in size. c. The added solute falls to the bottom of the container where it decreases in size for several hours and thereafter its size remains constant. d. The added solute causes the production of a large amount of solid white crystals.arrow_forwardWhat mass of a 4.00% NaOH solution by mass contains 15.0 g of NaOH?arrow_forwardDescribe the changes that occur between the time excess solute is placed into water and the time the solution becomes saturated.arrow_forward
- A chemist wishes to mix a solution that is12% acid. She has on hand 8 liters of a 8% acid solution and wishes to add some16% acid solution to obtain the desired 12% acid solution. How much 16% acid solution should she add? A chemist wishes to mix a solution that is 12% acid. She has on hand 6 liters of a 4% acid solution and wishes to add some 16% acid solution to obtain the desired 12% acid solution. How much 16% acid solution should she add?arrow_forwardClassify the substances by whether they are soluble or insoluble in water. Soluble in water Insoluble (or very low solubility) in water Answer Bank C,H12 (pentane, a nonpolar liquid) C12H„O1 (table sugar, a polar solid) KI (potassium iodide, an ionic solid) CH, (ethane, a nonpolar gas) CH,OH (methanol, a polar liquid)arrow_forwardPart1: 1.05×10-1 m glucose solution made by dissolving the glucose in 100.0 kg of water: ____ mol Part2: 2.35×10-2 m Na2CrO4 solution made by dissolving the Na2CrO4 in 1000.0 g of water:____ molarrow_forward
- A tank initially contains 200 liters of fresh water. Brine containing 2.5 N/liter of dissolved salt runs into the tank at the rate of 8 liters/min and the mixture kept uniform by stirring runs out at the same rate. After 15 minutes, what is the concentrations of the salt in the mixture? 1.1 N/L 1.8 N/L 2.1 N/L 0.82 N/Larrow_forward2. A beaker of water and a beaker containing a sugar solution were placed in a closed container at room temperature. After some time, the volume of water in the beaker decreased, while the volume of the sugar solution increased. Explain why. Time water sugar solution sugar water solutionarrow_forwardA solution is made by diluting 100 ml of a 0.15 mol/L standard solution to a volume of 0.150 L. What is the concentration of the resulting solution? 1.0.224 mol/L 2.0.158 mol/L 3.100 mol/L 4.0.1 mol/L 5.2.25 mol/Larrow_forward
- Calculate the molarity of a solution made by dissolving 0.382 mol glucose in enough water to make 1.32 L of solution. Your number should be followed by the correct symbol for molarity, but do not include "glucose" as part of your answer.arrow_forwardIn a certain medical test designed to measure carbohydrate tolerance, an adult drinks 7 ounces of a 30% glucose solution. When the test is administered to a child, the glucose concentration must be decreased to 20%. How much 30% glucose solution and how much water should be used to prepare 7 ounces of 20% glucose solution?arrow_forwardWhat volume of concentrated HCl solution contains 125 g of HCl?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





