
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 3P
Freshly prepared mitochondria were incubated with ß-hydroxybutyrate, oxidized cytochrome c, ADP, Pi, and cyanide. ß-hydroxybutyrate is oxidized by an NAD+ -dependent dehydrogenase. 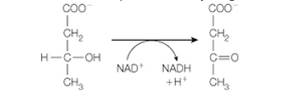
- The experimenter measured the rate of oxidation of ß-hydroxybutyrate and the rate of formation of ATP.
- Indicate the probable flow of electrons in this system.
- How many moles of ATP would you expect to be formed per mole of ß-hydroxybutyrate oxidized in this system?
- Why is ß-hydroxybutyrate added rather than NADH?
- What is the function of the cyanide?
- Write a balanced equation for the overall reaction occurring in this system (electron transport and ATP synthesis).
- Calculate the net standard free energy change (ΔGO') in this system, using
E'o values from Table 14.1 and a ΔGO' value for ATP hydrolysis of -32.2 kJ / mol.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Assume that a person was born with a defective NADH dehydrogenase complex, the complex that allows electron transport chain (ETC) to make ATP from NADH. Following consumption of a diet rich in carbohydrates and fats, would you expect this person to be able to produce any ATP by oxidative phosphorylation? Explain your answer?
Place the following components of the electron-transport chain in their proper order: (a) cytochrome c (b) Q-cytochrome coxidoreductase (c) NADH-Q reductase (d) cytochrome c oxidase (e) ubiquinone
Which of the following drives the electron transfer from coenzyme Q to cytochrome c, which also results to the pumping of protons from the mitochondrial matrix to the intermembrane space?
Complex I
Complex II
Complex III
Complex IV
If 2 molecules of FADH2 are oxidized during oxidative phosphorylation, how many molecules of ATP are synthesized?
Your answer should be a numerical input.
Chapter 14 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 14 - Prob. 1PCh. 14 - When pure reduced cytochrome c is added to...Ch. 14 - Freshly prepared mitochondria were incubated with...Ch. 14 - Prob. 4PCh. 14 - Prob. 5PCh. 14 - Prob. 6PCh. 14 - Intramitochondrial ATP concentrations are about 5...Ch. 14 - Prob. 8PCh. 14 - Prob. 9PCh. 14 - Years ago there was interest in using uncouplers...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- In the krebs cycle, if the enzyme used succinate dehydrogenase used NAD+ as the oxidizing agent, what impact would it have on the overall production of ATP in the call?arrow_forwardWhich of the following statements concerning the complete oxidation of FADH2 in the electron transport chain is NOT true? a. In the final step, electrons from cytochrome c to O2 reducing it to H2O in complex IV, and four protons are transported from the intermembrane space to the matrix. b. In the first step, electrons from FADH2 are transferred in complex II to ubiquinone, which does not transport any proton across the inner mitochondrion membrane. c. In the second step, complex III transfers the electrons from ubiquinone to cytochrome c, and four protons are transported from the matrix to the intermembrane space. d. The complete oxidation of FADH2 causes transfer of 6 protons and yields two ATP.arrow_forwardThe following statements describe the path of electrons from NADH through the electron transport chain. Please arrange them in order: Cytochrome c (Fe2+) carries electrons to Complex IV. Cytochrome c (Fe3+) is reduced to cytochrome c (Fe2+) QH2 carries electrons to Complex III Oxygen is reduced to water Ubiquinone (coenzyme Q) is reduced to ubiquinol (QH2)arrow_forward
- Which of the following statements is true? A. The NADH dehydrogenase complex can pump more protons than can the cytochrome b-c1 complex. B. The pH in the mitochondrial matrix is higher than the pH in the intermembrane space. C. The proton concentration gradient and the membrane potential across the inner mitochondrial membrane tend to work against each other in driving protons from the intermembrane space into the matrix. D. The difference in proton concentration across the inner mitochondrial membrane has a much larger effect than the membrane potential on the total proton-motive force.arrow_forwardAssume that 2.5 ATPs are generated per NADH and 1.5 ATPs per FADH2. How many ATPs are generated from the FADH2 and NADH molecules from each repetition of the ββbeta-oxidation pathway? Express your answer as an integer.arrow_forwardUncoupling agents are proteins spanning the inner mitochondrial membrane that allow protons to pass through the membrane and bypass the channel of ATP synthase. Describe the consequences to the proton gradient and ATP production.?arrow_forward
- Which complex of the mitochondrial electron transport chain is the only one able to accept electrons directly from FADH2? NADH dehydrogenase (complex I) cytochrome b-c1 (complex III) cytochrome c oxidase (complex IV) the ATP synthase complex FADH2 dehydrogenase (complex II) The above reaction (FADH2 donating its electrons to the electron transport chain) takes place in which of the following eukaryotic cell locations? the mitochondrial matrix the thylakoid membrane of chloroplasts the chloroplast stroma the inner mitochondrial membrane the cytoplasmarrow_forwardThe TCA cycle enzyme aconitase and the electron transport proteins have chromophores that absorb in the visible part of the spectrum, and therefore mitochondria have a dark brown color. In contrast, none of the glycolytic enzymes have visible spectrum chromophores. Which of the following statements is true? Question 3 Select one: a. Migrating birds, such as ducks have “white” breast meat, because flying long distances requires anaerobic ATP generation. d. The leg muscle of turkeys is specialized for anaerobic ATP generation via glycolysis. e. The flight muscle of turkeys is specialized for anaerobic ATP generation via glycolysis. f. None of the answers are correct.arrow_forwardIf 10 molecules of NADH are oxidized during oxidative phosphorylation, how many molecules of ATP are synthesized? Your answer should be a numerical input. Which of the following catalyzes the reduction of oxygen by using the electrons released from oxidizing coenzyme Q to reduce cytochrome c? NADH dehydrogenase Succinate dehydrogenase Cytochrome c reductase Cytochrome c oxidase Question 18 1 Point During glycolysis, the energy-investment phase occurs in the cytosol while the energy payoff phase occurs in the mitochondrial matrix. True Falsearrow_forward
- Consider the steps of the krebs cycle and electron carrier chain and answer: a) How many ATPs are produced from 10 Pyruvato molecules? b) How many ATPs are produced from 12 Acetyl CoA molecules?arrow_forwardIf 12 molecules of glucose enter the glycolytic pathway, how many ATP molecules can be generated in the electron transport chain from the NADH produced in the pathway assuming that there are 10 c subunits in the Fo complex of the organism?arrow_forwardYou know that during oxidative phosphorylation, 1 NADH creates 2.5 ATP and 1 FADH2 creates 1.5 ATP. From this information, how many total ATP molecules will be produced from the complete oxidation of pyruvate?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Mitochondrial mutations; Author: Useful Genetics;https://www.youtube.com/watch?v=GvgXe-3RJeU;License: CC-BY