
Concept explainers
(a)
Interpretation:
The structure of the alkyl bromide of molecular formula
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image.
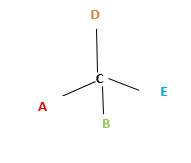
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different
(b)
Interpretation:
The structure of the
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image.
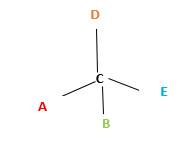
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different functional groups.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Label each compound as chiral or achiral. Compounds that contain a single carbon common to two rings are called spiro compounds. Because carbon is tetrahedral, the two rings are perpendicular to each other.arrow_forwardA compound that contains two different stereocenters can exist as four optically active stereoisomers. The flying wedge representations of the four stereoisomers are given below: ÇOOH ÇOOH ÇOOH ÇOOH H-C-OH HO-C-H H-C-OH HO-C-H H-C-OH HO-C-H HO-C-H H-C-OH ČH3 ČH3 ČH3 ČH3 II II IV What is the relationship between: 96.I and II? 97. I and IV? 98. I and III? II and III? III and IV? II and IV?arrow_forwardExplain the concept of chirality in organic chemistry, its significance in drug design, and the difference between enantiomers and diastereomers.arrow_forward
- < Question 12 of 67 Bystolic (shown below) is a pharmaceutical used to treat high blood pressure. What is the maximum number of stereoisomers possible for this structure? OH OH 1 4 7 +/- 10 stereoisomers 2 5 8 3 6 9 0 C x 100arrow_forwarda) HO OH Molecule 5 is achiral has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer b) Molecule 6 is achiral has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer Ph Molecule 7 is achiral has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomerarrow_forwardIdentify the relationship between the following two structures. OH A enantiomers B identical C OH C) neitherarrow_forward
- How many the chirality center(s) can you see in the following compound. 02 none H3C. 03 01 OH Br НО CH3arrow_forwardCount the chirality centers in each molecule below. CHO CH, ndete O 4 chiral centers O3 chiral centers O1 hiral centerarrow_forwardConsider the following pairs of structures. Identify the relationship between them by describing them as representing enantiomers, diastereomers, constitutional isomers, or two molecules of the same compound. H H3C" CH3 Br Hitm Br enantiomers constitutional isomers diastereomers H B H F F two molecules of the same compound CH3 H Ba CH3arrow_forward
- A) ОН B) HO. Molecule A is achiral and molecule B is chiral Molecule A is chiral and molecule B is achiral Both molecules are achiral. Both molecules are chiral.arrow_forwardPart B Identify the chiral carbon (s) in each of the following naturally occurring compounds, by stating the letter corresponding to the chiral carbon atom. 1. Amphetamine (Benzedrine), a stimulant used in treatment of hyperactivity. A CH3 CH₂CHNH2₂2 с 2. Norepinephrine, increases blood pressure and nerve transmission. HO B HO OH CB CHCH,NH, Aarrow_forwardIdentify the relationship between these compounds. OH NH₂ 요 H3C H HẠN H Ọ H CH3 Diastereomers Identical Enantiomers HO OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





