
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781305780170
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15.SE, Problem 54AP
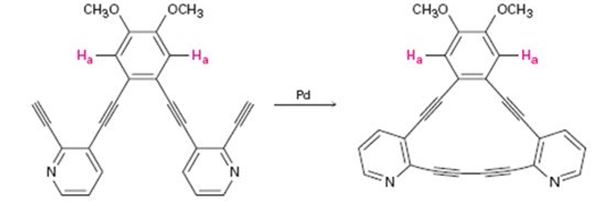
After the reaction below, the chemical shift of Ha moves downfield from 6.98 ppm to 7.30 ppm. Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A student planned to utilize HPLC to separate her chemically synthesized product (1) from th reactants (2 and 3).
Predict the elution sequence for compounds 1, 2 and 3 based on the above information. Justify youranswer.
How can we use UV/VIS spectroscopy to measure the rate of a reaction?
(UV spectroscopy lab, organic chemistry 2) Acetaldehyde shows two UV bands, one with a λmax of 289 nm (ε = 12) and one with a λmax of 182 nm (ε = 10,000). Which one is the n -> π* transition and which is the π -> π* transition? Explain your reasoning.
Chapter 15 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
Ch. 15.1 - Prob. 1PCh. 15.1 - Give IUPAC names for the following compounds:Ch. 15.1 - Prob. 3PCh. 15.2 - Pyridine is a flat, hexagonal molecule with bond...Ch. 15.3 - Prob. 5PCh. 15.4 - Draw the five resonance structures of the...Ch. 15.4 - Prob. 7PCh. 15.4 - Prob. 8PCh. 15.5 - Prob. 9PCh. 15.5 - Prob. 10P
Ch. 15.6 - Prob. 11PCh. 15.6 - How many electrons does each of the four nitrogen...Ch. 15.SE - Give IUPAC names for the following substances (red...Ch. 15.SE - All-cis cyclodecapentaene is a stable molecule...Ch. 15.SE - 1, 6-Methanonaphthalene has an interesting 1H NMR...Ch. 15.SE - Prob. 16VCCh. 15.SE - Azulene, an isomer of naphthalene, has a...Ch. 15.SE - Give IUPAC names for the following compounds:Ch. 15.SE - Draw structures corresponding to the following...Ch. 15.SE - Prob. 20APCh. 15.SE - Prob. 21APCh. 15.SE - Draw and name all possible aromatic compounds with...Ch. 15.SE - Propose structures for aromatic hydrocarbons that...Ch. 15.SE - Look at the three resonance structures of...Ch. 15.SE - Prob. 25APCh. 15.SE - Prob. 26APCh. 15.SE - Look at the five resonance structures for...Ch. 15.SE - Prob. 28APCh. 15.SE - 3-Chlorocyclopropene, on treatment with AgBF4,...Ch. 15.SE - Prob. 30APCh. 15.SE - Prob. 31APCh. 15.SE - Prob. 32APCh. 15.SE - Which would you expect to be most stable,...Ch. 15.SE - How might you convert 1, 3, 5, 7-cyclononatetraene...Ch. 15.SE - Calicene, like azulene (Problem 15-17), has an...Ch. 15.SE - Pentalene is a most elusive molecule that has been...Ch. 15.SE - Prob. 37APCh. 15.SE - Prob. 38APCh. 15.SE - Compound A, C8H10, yields three substitution...Ch. 15.SE - Prob. 40APCh. 15.SE - Propose structures for compounds that fit the...Ch. 15.SE - Prob. 42APCh. 15.SE - Prob. 43APCh. 15.SE - N-Phenylsydnone, so-named because it was first...Ch. 15.SE - Prob. 45APCh. 15.SE - Prob. 46APCh. 15.SE - Prob. 47APCh. 15.SE - Propose a structure for a molecule C14H12 that has...Ch. 15.SE - The proton NMR spectrum for a compound with...Ch. 15.SE - The proton NMR spectrum of a compound with formula...Ch. 15.SE - Aromatic substitution reactions occur by addition...Ch. 15.SE - Prob. 52APCh. 15.SE - Consider the aromatic anions below and their...Ch. 15.SE - After the reaction below, the chemical shift of Ha...Ch. 15.SE - Prob. 55APCh. 15.SE - Azo dyes are the major source of artificial color...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why is it important for solutions to be diluted before they are run through spectroscopy?arrow_forward1a) Draw the keto-enol equilibrium reaction for the following ketone 1b) Figure 1 shows three IR spectra, labelled A, B and C. Identify which one corresponds to the ketone shown in section 1a). Justify your answer by discussing the appropriate bands of the three spectra.arrow_forward3. If you would directly analyse a solid sample with AAS, which method will you prefer? Explain about it in detail.arrow_forward
- hi, can someone please help me interpret this IR? it was done in the lab and I've also attached the reaction we did to obtain this IR. which peaks correspond to the product/reactant/impurities? Thanks!arrow_forwardHow would the equilibrium be affected in regards to the value of Qc or Kc for each of the following conditions?: Fe3+ (aq) + SCN- (aq) ⇌ FeSCN2+ (aq) Adding KSCN (aq) Adding Fe(NO3) (aq) Adding NaF (s) Placing the solution into a hot water bath Placing the solution into an ice bath (To whom this may concern, Could you at least explain one as simply as possible so I'm able to understand the others enough to explain them myself? Thanks in anvance!)arrow_forwardIf you put methylene blue and disperse red 1 mixture into salad dressing, predict what will you observearrow_forward
- Since the resolution of SrCO3 is a g/L, the resolution product of SrF2 is B, what happens [F -] when equilibrium is reached if solid SrF2 is boiled with a solution of Na2CO3 so that it is [CO32 -]= C Molar in the final solution?arrow_forwardPlease explain the number of signals and the integration that is observed in the 1H NMR spectrum for the major product of the reaction from question 6. - Focus on the alkyl protons, in particular those two methylene (CH2) groupsarrow_forwardconstruct the expression for Kc for the following reaction 3H2(g)+6C(s)=C6H6(g)arrow_forward
- Following is a 1H-NMR spectrum of 2-butanol. Explain why the CH2 protons appear as a complex multiplet rather than as a simple quintet.arrow_forwardAcetaldehyde shows two UV bands, one with a λmax of 289 nm (ε = 12) and one with a λmax of 182 nm (ε = 10,000). Which one is the n -> π* transition and which is the π -> π* transition? Explain your reasoning.arrow_forwardA student planned to utilize HPLC to separate her chemically synthesized product (1) from th reactants (2 and 3). Q1 - Which mode of separation would be reasonable to separate these compounds? Justify your answerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY