
Concept explainers
Interpretation:
The IR band in Figure 16-33 that indicates an
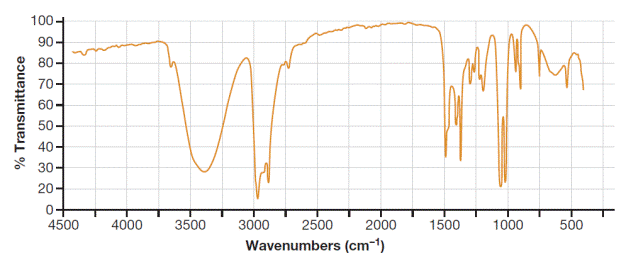

Concept introduction:
In general, the
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Using our Table of Infrared Absorbances, which functional group absorbs at 1,725, 2,720 and 2,820 cm-1 ? These three peaks are definitive/diagnostic/mean this group is really there. carboxylic acid amide amine aldehyde ketonearrow_forwardYou have an unknown with an absorption at 1680 cm-1; it might be an amide, an isolated double bond, a conjugated ketone, aconjugated aldehyde, or a conjugated carboxylic acid. Describe what spectral characteristics you would look for to help youdetermine which of these possible functional groups might be causing the 1680 peak.Sarrow_forward3-Bromo-1-phenyl-1-propene shows a complex NMR spectrum in which the vinylic proton at C2 is coupled with both the C1 vinylic proton (J = 16 Hz) and the C3 methylene protons (J = 8 Hz). Draw a tree diagram for the C2 proton signal, and account for the fact that a five-line multiplet is observed.arrow_forward
- You have an unknown with an absorption at 1680 cm-1; it might be an amide, an isolated double bond, a conjugated ketone, aconjugated aldehyde, or a conjugated carboxylic acid. Describe what spectral characteristics you would look for to help youdetermine which of these possible functional groups might be causing the 1680 peak.arrow_forwardLabel the 13C spectrum of (R,R)-N,N’-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine peaks and explain why they appear at the ppm that they show up at.arrow_forwardShow which carbon atoms correspond with which peaks in the 13C NMR spectrum ofbutan-2-onearrow_forward
- What functional group can you get from its IR spectrum? Support your answer by identifying prominent peaks (wavenumbers cm^-1) and assigning the functional group.arrow_forwardFill in the table by matching the numbers of the structure to their corresponding peaks of the C-13 spectra.arrow_forwardIdentify the Campesterol peak of the GC-MS of Burn Morel mushrooms (Tomentosa) and the M (+/-) peaks that are present. Include the mass and signature intensities of the peaks main product.arrow_forward
- These are four alcohols with the formula C4H10O. One of them produced this C-NMR spectrum, which alcohol did that and what carbons correspond with what peaks?arrow_forwardIdentify and explain the IR absorption characteristics of the unknown C6H14O in the 1400 to 4000 cm-1 range. Then identify the class of the unknown compound C6H14O. At the broad peak I believe it is either an alcohol & phenol or an amine. For the second peak i think it is an alkane.arrow_forwardIdentify the frequency of both carbonyl stretches in your IR. Which one corresponds to the benzyl ester? Wavenumber (cm-1) Bond Vibration Shape Intensity Functional Group 3288.63 cm-1 N-H Stretch Sharp Weak Amine/Amide 3078.39 cm-1 C-H Stretch Sharp Weak Aromatic/Alkene 2976.16 cm-1 C-H Stretch Sharp Weak Alkane 1720.50 cm-1 C=O Stretch Sharp Medium Ester 1653.00 cm-1 C=O Stretch Sharp Medium Amide 1556.55 cm-1 C=C Stretch Sharp Medium Aromaticarrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

