
Interpretation:
The structure of the compound of the formula
Concept introduction:
The structure of a compound can be determined from the IR and NMR spectra.
The IR spectrum of a molecule consists of several absorption peaks/bands. These bands, particularly those above about 1200
The
The
The formula of the compound, if known, can provide information about the unsaturation, number of double or triple bonds, and rings in the structure. This is known as the Index of Hydrogen Deficiency (IHD), and for a molecule containing only C, H, and O, it is calculated as
Using this information, the possible ways in which the fragments and functional groups are connected can be determined, leading to the probable structure of the molecule.
Answer to Problem 16.84P
The structure of the compound is
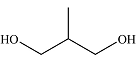
Explanation of Solution
The formula of the compound is
This possibly means the oxygen atoms are part of alcohol or ether groups.
The
The data from the spectrum can then be summarized as follows:
| δ ppm | No. of H’s | Splitting | Coupled H’s | Fragment |
| 0.85 | 3 | Doublet | 1 | 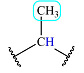 |
| 1.9 | 1 | Broad | - | 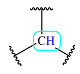 |
| 3.6 | 4 | Doublet | 1 | 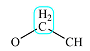 |
| 4.1 | 2 | Triplet | 2 | OH |
The fragments can then be put together as

The presence of the alcoholic OH group is supported by the broad IR absorption band at about 3500
The structure of a compound was determined from the
Want to see more full solutions like this?
Chapter 16 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





