
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.32UKC
One phosphorylated form of glycerate is 3-phosphoglycerate (a
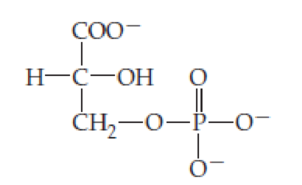
- (a) Identify the type of linkage between glycerate and phosphate.
- (b) 1,3-Bisphosphoglycerate (two phosphates on glycerate) has an anhydride linkage between the carbonyl at C1 of glycerate and phosphate. Draw the structure of 1,3-bisphosphoglycerate (another metabolic intermediate).
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(b)
Biotin is a coenzyme required for a key step in gluconeogenesis.
(i) To which enzyme is biotin linked and what is the nature of the bond?
(ii) What properties do the biotin and the linkage have that enable biotin to interact with two distinct
active sites in this enzyme?
(iii) What chemical steps take place at each of these active sites (no need to specify the complete
reactions)?
Which of an alpha-D-2,3-di-O-methylglucopyranose or alpha-D-2,3, 6-tri-O-methylglucopyranose represents a glucose unit in glycogen which was originally carrying an (alpha 1-->6) glycosidic bond?
The conversion of 1,3-bisphosphoglycerate (1,3-BPG) to glyceraldehyde 3-phosphate (GAP) represents
a reduction.
an oxidation.
no change in oxidation state.
d reaction is
Chapter 17 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 17.1 - Identify the following molecules as a carboxylic...Ch. 17.1 - Prob. 17.2PCh. 17.1 - Prob. 17.3PCh. 17.1 - Prob. 17.4PCh. 17.1 - Prob. 17.5PCh. 17.1 - Prob. 17.6PCh. 17.1 - In the following pairs of compounds, which would...Ch. 17.1 - Write both condensed and line structures for (a)...Ch. 17.1 - Prob. 17.9PCh. 17.1 - Draw structures corresponding to these names: (a)...
Ch. 17.1 - Prob. 17.11PCh. 17.1 - Prob. 17.12PCh. 17.1 - Prob. 17.13KCPCh. 17.2 - Salsalate, which is an ester formed by the...Ch. 17.2 - Prob. 17.2CIAPCh. 17.2 - Prob. 17.3CIAPCh. 17.2 - Prob. 17.14PCh. 17.2 - Prob. 17.15PCh. 17.2 - Prob. 17.16PCh. 17.3 - Prob. 17.17PCh. 17.3 - Raspberry oil contains an ester that is made by...Ch. 17.3 - Prob. 17.19PCh. 17.3 - Prob. 17.20PCh. 17.3 - Prob. 17.21PCh. 17.4 - If a bottle of aspirin tablets has the aroma of...Ch. 17.4 - Prob. 17.23PCh. 17.4 - What carboxylic acids and amines result from...Ch. 17.5 - Prob. 17.25PCh. 17.5 - Prob. 17.26KCPCh. 17.6 - Prob. 17.27PCh. 17.6 - Prob. 17.28PCh. 17.6 - Prob. 17.4CIAPCh. 17.6 - Prob. 17.5CIAPCh. 17.6 - Prob. 17.29PCh. 17 - Prob. 17.30UKCCh. 17 - Prob. 17.31UKCCh. 17 - One phosphorylated form of glycerate is...Ch. 17 - Prob. 17.33UKCCh. 17 - Prob. 17.34UKCCh. 17 - Prob. 17.35UKCCh. 17 - Prob. 17.36UKCCh. 17 - For the following compounds, give the systematic...Ch. 17 - Write the equation for the ionization of hexanoic...Ch. 17 - Prob. 17.39APCh. 17 - Prob. 17.40APCh. 17 - Prob. 17.41APCh. 17 - Give systematic names for the following carboxylic...Ch. 17 - Give systematic names for the following carboxylic...Ch. 17 - Prob. 17.44APCh. 17 - Prob. 17.45APCh. 17 - Draw structures corresponding to the following...Ch. 17 - Draw structures corresponding to the following...Ch. 17 - Malic acid, a dicarboxylic acid found in apples,...Ch. 17 - Prob. 17.49APCh. 17 - Prob. 17.50APCh. 17 - Prob. 17.51APCh. 17 - Prob. 17.52APCh. 17 - Prob. 17.53APCh. 17 - Give systematic names for the following structures...Ch. 17 - Give systematic names for the following structures...Ch. 17 - Prob. 17.56APCh. 17 - Prob. 17.57APCh. 17 - Give systematic names for the following structures...Ch. 17 - Give systematic names for the following structures...Ch. 17 - Prob. 17.60APCh. 17 - What compounds are produced from hydrolysis of...Ch. 17 - Procaine, a local anesthetic whose hydrochloride...Ch. 17 - Prob. 17.63APCh. 17 - Lactones are cyclic esters in which the carboxylic...Ch. 17 - When both the carboxylic acid and the amine are in...Ch. 17 - LSD (lysergic acid diethylamide), a semisynthetic...Ch. 17 - Prob. 17.67APCh. 17 - Prob. 17.68APCh. 17 - Prob. 17.69APCh. 17 - Prob. 17.70APCh. 17 - Prob. 17.71APCh. 17 - Prob. 17.72APCh. 17 - Prob. 17.73APCh. 17 - Prob. 17.74APCh. 17 - Prob. 17.75APCh. 17 - Three amide isomers, N,N-dimethylformamide,...Ch. 17 - Prob. 17.77CPCh. 17 - Prob. 17.78CPCh. 17 - Mention at least two simple chemical tests by...Ch. 17 - Prob. 17.80CPCh. 17 - Name the following compounds.Ch. 17 - Each of the following materials has an ester that...Ch. 17 - Prob. 17.83GPCh. 17 - Prob. 17.84GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- a) Draw an alpha 16 linkage between N-acetylglucosamine (phosphorylated at carbon number 6) and a-glucose-1-phosphate. b) Will this disaccharide contribute to the formation of a precipitate in a solution of alkaline copper? Very briefly explain your answer. (arrow_forwardName and draw the structure of the a-keto acid resulting when each of the following amino acids undergoes transamination with a-ketoglutarate: (a) aspartate, (b) glutamate, (c) alanine, (d) phenylalanine.arrow_forwardLithium ion inhibits the synthesis of inositol trisphosphate by inhibiting a reaction in the breakdown of inositol trisphosphate.Explain this apparent paradox.arrow_forward
- Lithium ion inhibits the synthesis of inositol trisphosphate by inhibiting a reaction in the breakdown of inositol trisphosphate. Explain this apparent paradox.arrow_forwardWhile fatty acids longer than 20 carbons are rarely found in foods, lignoceric acid (24:0) is found in a variety of tree nuts. Answer the following based on the conversion of a molecule of lignoceric acid to 8-hydroxybutyrate. (a) What are the 8-oxidation products and how many ATP are required during activation for one molecule of lignoceric acid? (b) Given the following, how many molecules of 8-hydroxybutyrate can be produced? CoA 2 2 CoA NADH NAD+ H+ OH ẞ-hydroxybutyrate (c) Based on the total NADH and FADH2 available after converting lignoceric acid into 8-hydroxybutyrate, what is the maximum yield of ATP that can be produced in the liver? Don't forget to include any ATP required for activation steps.arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forward
- Acetone, CH3–(C=0) –CH3, and urea, NH2–(C=0)–NH2, have similar chemical structures. (a) Compare the possible intermolecular forces for acetone with those for urea. (b) Compare the intermolecular forces between phycocyanin and acetone with those between phycocyanin and water. Briefly explain how acetone disrupts the folding in phycocyanin. (c) Compare the intermolecular forces between phycocyanin and urea with those between phycocyanin and water. Briefly explain how urea disrupts the folding in phycocyanin. (d) Compare your experimental observation on the disruption of phycocyanin folding in the presence of acetone with that in the presence of urea. Briefly explain whether your comparison is consistent with the properties of acetone and urea.arrow_forward11.32) Identify the following as properties of either glycogen, amylopectin, both glycogen and amylopectin, or neither glycogen nor amylopectin. a) contains a-(1-6) glycosidic bonds both glycogen and a my lopectin b) contains ß-(1→6) glycosidic bonds neither glycogen nor amylopectin c) contains ß-(1→4) glycosidic bonds neither glycogen nor a mylopectin d) contains glucose and fructose residues only neither glycogen nor amylopectin e) homopolysaccharide both glycogen and a my lo pectin f) heteropolysaccharide neither glycogen nor a mylopectin g) branching occurs less frequently (glycogen or amylopectin) amy lo pectin h) contains helical structures both glycogen and amylopection i) found in plants amy lopectinarrow_forwardCeruloplasmin is a blue-colored monomeric oxidase found in mamma- lian blood plasma. It contains multiple copper atoms per mole- cule. A ribbon representation of its three-dimensional shape is shown below: a) b) c) Identify any cofactor(s) which ceruloplasmin may possess. How do you know? Does ceruloplasmin act as an enzyme ? Describe the secondary, tertiary and quaternary structures of ceruloplasmin.arrow_forward
- This is a conjectural question: If the reactive part of coenzyme A is the thioester, why is the molecule socomplicated?arrow_forwardHuman blood serum contains a class of enzymes known as acid phosphatases, which hydrolyze biological phosphate esters under slightly acidic conditions (pH 5.0): R-O-P-O3-2 + H2O --> R-OH + HO-P-O3-2. Acid phosphatases are produced by erythrocytes, the liver, kidney, spleen, and prostate gland. The enzyme from the prostate gland is clinically important because an increased activity in the blood is frequently an indication of cancer of the prostate gland. The phosphatase from the prostate gland is strongly inhibited by tartrate ion, but acid phosphatases from other tissues are not. How can this information be used to develop a commercial specific procedure for measuring the activity of the acid phosphatase of the prostate gland in human blood serum? * 1. Prostate cancer cannot be diagnosed biochemically. 2. Use tartrate to inhibit phosphatase from prostate gland and then subtract the results from the total serum enzyme activities to get an…arrow_forwardThe free energy of hydrolysis of an α(1→4) glycosidic bond is −15.5 kJ ⋅mol−1, whereas that of an α(1→6) glycosidic bond is −7.1 kJ ⋅ mol−1. Use these data to explain why glycogen debranching includes three reactions [breaking and re-forming α(1→4) bonds and hydrolyzing α(1→6) bonds], whereas glycogen branching requires only two reactions [breaking α(1→4) bonds and forming α(1→6) bonds].arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Metabolic Pathways; Author: Wisc-Online;https://www.youtube.com/watch?v=m61bQYio9ys;License: Standard Youtube License