
Interpretation:
The functional groups present in lidocaine has to be identified. Also the structure of
Concept Introduction:
Carboxylic acid: One
Amide: One
Amide Formation: Amide is formed when a carboxylic acid reacts with an amine or ammonia.
- Primary amide is produce when a carboxylic acid reacts with ammonia.
- Secondary and tertiary amide is produce when a carboxylic acid reacts with primary and secondary amine respectively.
- Ammonium salt is formed when tertiary amine and a carboxylic acid reacts forming an ionic compound with a carboxylate acid anion and a trialkyl ammonium cation since there is no hydrogen atom in trialkyl amine
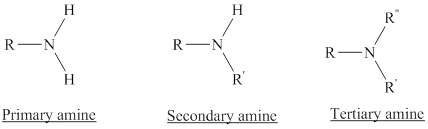
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Aromatic Compounds: Compounds that are planar, cyclic and having
Trending nowThis is a popular solution!

Chapter 17 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Explain what indole alkaloids are. And describe the significance of vinflunine.arrow_forwardFumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forwardWhat makes 2, 4-dinitrophenylhydrazine suitable for characterizing aldehydes and ketones? How would carbonyl groups in the two pheromones be distinguished by a suitable chemical method?arrow_forward
- Methadone is dispensed as its hydrochloride salt. What is the use of the salt rather than the amine?arrow_forwardGive the pharmacological property of the following alkaloids. Where do they naturally occur? morphine quinine berberine What are the chemical tests used in identifying alkaloids?arrow_forwardWhy is salbutamol better than isoproterenol in asthma management in terms of their chemical structure?arrow_forward
- Name at least two undesirable characteristics are often associated with alkaloids.arrow_forward94arrow_forwardThe analgesic phenacetin is synthesized by treating 4- ethoxyaniline with acetic anhydride. a. Which of the following is the structure of 4- ethoxyaniline? OCH₂CH3 NH₂ OCH₂CH3 NH₂ OCH₂CH3arrow_forward
- 2-butanol can be formed as the only product of the Markovnikov addition of H2O to two different alkenes. In contrast, 2-pentanol can be formed as the only product of the Markovnikov addition of H2O to just one alkene. To examine the difference, draw the alkene starting materials of each alcohol. : Draw the bond-line (skeletal) structures of the two alkene starting materials that can be used to synthesize 2-butanol via Markovnikov hydration. Part 1 of 2 Click and drag to start drawing a structure. ☑arrow_forwardWhat is the ionisable group of phenazopyridine? With illustrationsarrow_forwardHow aspirin is prepared from salicylic acid?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





