
EBK LIFE: THE SCIENCE OF BIOLOGY
11th Edition
ISBN: 8220103935432
Author: Sadava
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 17, Problem 3Q
Summary Introduction
To review:
The level of activity of the enzyme coded by ‘A’ allele compared to the enzyme coded by ‘G’ allele.
Given:
In the given experiment, the rate of
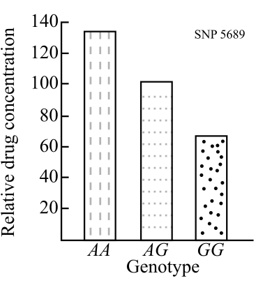
Introduction:
The metabolism of drug is effective in people with genotype GG where drug concentration in the blood is around 70. The drug concentration for the genotype AG is around 100 and it is around 130 for AA genotype. It is assumed that the SNP 5689 is present in a gene, which encodes an enzyme. This enzyme breaks down the drug.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the mechanism of the enzyme RNase:
What would happen to the Km (i.e., would it increase, decrease, or stay the same) if the his12 was mutated to a lysine? Explain.
What would happen to the Kcat (i.e., would it increase, decrease, or stay the same) if the his12 was mutated to a valine? Explain.
Consider a mutant form of the lac repressor protein whose ΔGo for binding DNA differs from that of the wild-type protein by 2 kcal/mol, the approximate free energy equivalent of a single hydrogen bond. What is the difference (i.e., the ratio) of the Kd values (for DNA binding) of the wild-type versus the mutant lac repressor protein?
You generate mutants in the metabolic pathway for starlase. You conduct some complementation
tests (after testing for dominance of course) and come up with the following results:
1
2
4
6
1
+
+
+
2
+
3
+
4
+
5
6
a. How many complementation groups are there? [Select]
b. You conduct some additional experiments to elucidate the starlase metabolic pathway. Your
results are shown below. Use this information alongside information from the complementation
table above to place the intermediates in the correct order on the pathway. (HINT: use the
complementation groups from the table above to help you consolidate information on the tables
below. Reference practice question 3 from today's lecture for help).
Addition to minimal medium
Mutant
None
starlase
P
1
+
+
2
+
+
4
+
+
5
+
6
Precursor -->
[ Select ]
[ Select ]
[ Select ]
--> starlase
c. Mutant 4 has a loss-of-function mutation for which enzyme in the starlase synthesis pathway?
[ Select ]
E1
E2
ЕЗ
Е4
Precursor > Intermediate 1→ Intermediate…
Chapter 17 Solutions
EBK LIFE: THE SCIENCE OF BIOLOGY
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Based on the information in as shown, which single-nucleotide mutationevent is more likely: Arg-to-His, or Arg-to-Ser? Explain.arrow_forwardIn the table below, there are four versions of gene A, one of which is normal, and the other three which contain mutations that make the gene product nonfunctional. Focus on the shaded region of the sequence. Use the genetic code table to answer the question. How would you describe Mutation #2? Partial DNA sequence for gene A ("..." indicates many nucleotides of sequence not shown) 5' ... ATG GTG AGC AAG GAG GAG CTG TTC ACC TGT AAA TAG ... Normal Mutation #1 5' ... ATG GTG AGC AAG GAG AAG CTG TTC ACC TGT AAA TAG ... Mutation #2 5' ... ATG GTG AGC AAG TAG GAG CTG TTC ACC TGT AAA TAG ... Mutation #3 5' ... ATG GTG AGC AAG GAG CTG TTC ACC TGT AAA TAG ... Silent mutation Nonsense mutation Frameshift mutations Missense mútationarrow_forwardThis shows where EcoR1 cuts, where HindIII cuts, and where BstEII cuts. Given that the entire chromosome is 48,502 base pairs (as it says), figure out the fragment sizes you will expect if you cut it with EcoR1, or with HindIII, or with BseEII. Draw a map like the example attached " Show the sizes of each piece. Color code it according to which enzyme cut it.arrow_forward
- A site-directed mutagenesis experiment was done on the catalytic triad of the serine protease subtilisin where all of the amino acids of the catalytic triad were mutated to alanine. The triple mutant enzyme was characterized kinetically and the mutant displayed a thousand-fold (103) rate enhancement over the uncatalyzed reaction. Explain the source of this rate enhancement. (The native wild-type subtilisin has a rate acceleration of 1010, when compared to the uncatalyzed reaction.)arrow_forwardAn experiment was conducted with two groups of genetically identical female agouti mice (Avy/a) that were mated with normal (a/a) male mice . Both female groups ate lab chow with bisphenol A (BPA) (50mg/Kg). (BPA, used in the manufacture of polycarbonate plastics, has estrogenic effects that include decreased CpG methylation.) The lab chow of one group was supplemented with methyl donors (e.g., folate, choline, and vitamin B12). The effects of BPA were abolished in the Avya offspring of the mice fed with the methyl donor–supplemented chow (i.e., the coat color distribution observed was shifted toward brown). Explain.arrow_forwardThe following is a list of mutational changes. For eachof the specific mutations described, indicate which ofthe terms in the right-hand column applies, either as adescription of the mutation or as a possible cause.More than one term from the right column can applyto each statement in the left column.1. an A–T base pair in the wild-type gene ischanged to a G–C pair2. an A–T base pair is changed to a T–A pair3. the sequence AAGCTTATCG is changed toAAGCTATCG4. the sequence CAGCAGCAGCAGCAGCAGis changed toCAGCAGCAGCAGCAGCAGCAGCAG5. the sequence AACGTTATCG is changed toAATGTTATCG6. the sequence AACGTCACACACACATCGis changed to AACGTCACATCG7. the sequence AAGCTTATCG is changed toAAGCTTTATCGa. transitionb. basesubstitutionc. transversiond. deletione. insertionf. deaminationg. X-rayirradiationh. intercalatori. slippedmispairingarrow_forward
- You are interested in studying a unique protein found in a rare wildflower found in the desert region of Atacama. The wild-type amino acid sequence isolated from this flower is: Wildtype: Arg-Lys-Thr-Leu-Gly-Arg A mutant for the gene that specifies this protein is isolated and the amino acid sequence of its protein is determined: Mutant: Arg-Lys-Thr-Leu-Gly-Gly a) Identity what the mutation changed in the amino acid sequence b) What is the effect of the mutation at the amino acid level? c) What is the effect of the mutation at the RNA level? d) What kind of mutation happened at the DNA level?arrow_forwardRecall that Leber congenital amaurosis (LCA), a formof congenital blindness in humans, can be caused byhomozygosity for recessive mutations in the RPE65gene. Recently, a rare dominant mutation in RPE65has been implicated as one cause of an eye diseasecalled retinitis pigmentosa, which is characterized byretinal degeneration that can progress to blindness.The dominant RPE65 mutation is a missense mutation causing amino acid 447 in the polypeptide tochange from Asp to Glu. Little is known about thenature of the mutant protein.a. Do you think that the dominant allele is morelikely a loss-of-function or a gain-of-functionmutation? Explain.b. As described in this chapter, gene therapy for LCAhas been at least partially successful. Do you thinkthat the same kind of gene therapy can be used forpatients with retinitis pigmentosa caused by thedominant mutant allele of RPE65? Explain.arrow_forwardWhat are histone deacetylase inhibitors (HDACi)? Give example(s)arrow_forward
- For the genotypes and conditions (lactose present or absent) shown in the following Table 2, predict whether the expression of beta-galactosidase is inducible (I), noninducible (N) or constitutive (C). Explain your reason. Table 2 Genotype I*p*O°z* Condition (i) No lactose (ii) I*P*O*Z+ Lactose I*P-0*Z* (iii) (iv) Lactose ISP+O°Z+ Lactose (v) Ip*o°Z+ No lactosearrow_forwardYou would like to add a nuclear localization sequence (NLS) of Lys-Lys-Lys-Arg-Lys to a protein that is usually found in the cytoplasm of a yeast cell. To accomplish this, you introduce the nucleotide sequence encoding the NLS into the gene that encodes the cytoplasmic protein of interest. a. What is the size of the nucleotide insert that will encode the NLS? Briefly explain. 5' 3' b. Below is a diagram of the gene encoding the cytoplasmic protein of interest in the yeast genome. If your goal is to put the NLS at the carboxyl (C) terminus of the protein, at which location (A-E) should the NLS be inserted? Briefly explain. A TATAA ATATT promoter +1 B ATG TAC D TAA ATT stop codon E 3' 5'arrow_forwardWhat can you determine from the r-value about the relationship between gene copy number and amylase enzyme concentration? r value = 0.935833048arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Mitochondrial mutations; Author: Useful Genetics;https://www.youtube.com/watch?v=GvgXe-3RJeU;License: CC-BY