
Concept explainers
Epoxide Rearrangements and the NIH Shift
This passage is about two seemingly unrelated aspects of
epoxide rearrangements
arene oxides
These two topics merge in an important biological transformation in which neither the
reactant nor the product is an epoxide

Epoxide rearrangements
In some epoxide ring-opening reactions
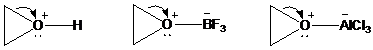
As positive charge develops on the ring carbon, one of the groups on the adjacent carbon migrates to it. This migration is assisted by electron
all of this occurs in the same transition state. Subsequent deprotonation gives an
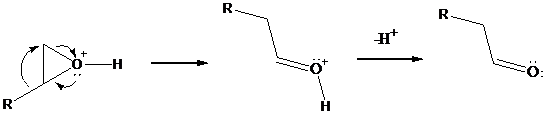
Overall, the reaction resembles the pinacol rearrangement of vicinal
Descriptive Passage and Interpretive Problems) and takes place under similar conditions.
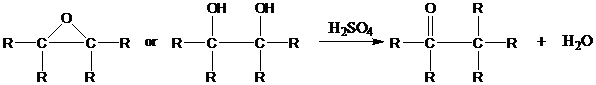
Arene Oxides
Aromatic rings are normally inert to the customary reagents that convert
arene oxides have been synthesized in the laboratory, often by indirect methods. Their chemical
reactivity resembles that of other epoxides.

The most striking thing about arene oxides is their involvement in biological processes. Enzymes
In the liver oxidize
The NIH shift
Although hydroxylation of phenylalanine to tyrosine looks like a typical electrophilic aromatic sub stitution, scientists at the U.S. National Institutes of Health discovered that the biochemical pathway combines epoxidation of the benzene ring followed by epoxide ring opening with rearrangement. This rearrangement, which is the biochemical analog of the pinacol

Acetanilide, which has pain-relieving properties, undergoes a biochemical oxidation
similar to that of the NIH shift that occurs with phenylalanine. The product formed from
acetanilide is itself a pain reliever. What is the structure of this substance (better known as
Tylenol)?
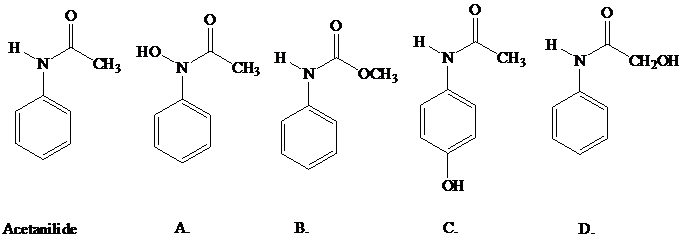
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
ORGANIC CHEMISTRY (LL)-W/SOLN.>CUSTOM<
- A newer generation of antipsychotics, among them clozapine, are now used to treat the symptoms of schizophrenia. These drugs are more effective than earlier drugs in improving patient response in the areas of social withdrawal, apathy, memory, comprehension, and judgment. They also produce fewer side effects such as seizures and tardive dyskinesia (involuntary body movements). In the following synthesis of clozapine, Step 1 is an Ullmann coupling, a type of nucleophilic aromatic substitution that uses a copper catalyst. (a) Show how you might bring about formation of the amide in Step 2. (b) Propose a reagent for Step 3. (c) Propose a mechanism for Step 4. (d) Is clozapine chiral? If so, how many of the possible stereoisomers are formed in this synthesis?arrow_forwardAll rearrangements we have discussed so far have involved generation of an electron-deficient carbon followed by a 1,2-shift of an atom or a group of atoms from an adjacent atom to the electron-deficient carbon. Rearrangements by a 1,2-shift can also occur following the generation of an electron-deficient oxygen. Propose a mechanism for the acid-catalyzed rearrangement of cumene hydroperoxide to phenol and acetone.arrow_forwardThe following reaction involves two sequential Heck reactions. Draw structural formu- las for each organopalladium intermediate formed in the sequence and show how the final product is formed. Note from the molecular formula given under each structural formula that this conversion corresponds to a loss of H and I from the starting material. Acetonitrile, CH,CN, is the solvent. 1% mol Pd(OAc), 4% mol Ph,P CH,CN C4H171 C4H16arrow_forward
- Predict the b-elimination product(s) formed when each bromoalkane is treated with sodium ethoxide in ethanol. If two or more products might be formed, predict which is the major productarrow_forwarduo 9:0A Asiacell I. 02:48 المتبقي متعد د الخيارات Which alkyl halide would react the fastest under substitution conditions with HO-? 2-bromo-2-methylpentane O 2-bromo-3-methylpentane 1-bromo-2,2-dimethylpentane 2-bromopentane 1-bromo-4-methylpentane O 1 of 5arrow_forward#16c. Provide the missing reactants, reagents, or products for the following reaction sequences below.arrow_forward
- Aldehydes undergo nucleophilic addition reactions with potent nucleophiles such as water, alcohol and amino compounds. Which of the following nucleophiles will exhibit the greatest ability to attack the electrophilic carbon in the carbonyl group of ethanal? cyclohexyl alcohol para-chlorophenol isoamyl alcohol ortho-aminophenol An aromatic ring should satisfy Huckel’s rule, wherein the number of electrons participating in the cyclic conjugation should be equal to 4n + 2. Which of the following cyclic structures does NOT obey Huckel’s rule? Cyclobutadienyl dianion Tetrahydrofuran ring Cyclopropene structure Pyrimidine ring structure What is the role of H • ions in the nucleophilic substitution of alcohols usinghydrogen halides? Removal of an alkyl groupProtonation of -OH groupStabilization of carbocationActivation of oxygen radicalarrow_forwardIn the acid-catalyzed dehydration of 2-methyl-1-propanol, what carbocation would be formed if a hydride shift accompanied cleavage of the carbon–oxygen bond in the alkyloxonium ion? What ion would be formed as a result of a methyl shift? Which pathway do you think will predominate, a hydride shift or a methyl shift?arrow_forward1) Nucleophilic substitution reaction of alkyl halide is a process when nucleophile replace the leaving group of alkyl halide. Propose the mechanism and product(s) for the following substitution reaction.arrow_forward
- Denote how many nucleophilic centers are present in the following molecular structure (in case of polar bonds show clearly on the structure the dipoles õ+ and d-). :o: сноarrow_forwardExplain the relevance pi* (antibonding) -molecular orbitals have in nucleophilic addition reactions of carbonyl containing compounds.arrow_forwardKetones and aldehydes react with sodium acetylide (the sodium salt of acetylene) to give alcohols, as shown in the following example: R1 ОН 1. НС—с: Na* R2 R1 R2 2. Нзо" HC Draw the structure of the major reaction product when the following compound reacts with sodium acetylide, assuming that the reaction takes preferentially from the Si face of the carbonyl group. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • You do not have to explicitly draw H atoms. • If a group is achiral, do not use wedged or hashed bonds on it. CH3 CHarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

