
Concept explainers
(a)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
The normal reaction of an amide with water does not give good yield of product. As amides are least reactive carboxylic acid derivatives so to increase the reactivity of amides a mineral acid is added. The addition of acid increases the reactivity of amide by making more electrophilic carbonyl carbon and it also helps in the elimination of amine part from amide via protonating the amine group during reaction and make it a good leaving group.
The reaction of water and amides in presence of acid gives a carboxylic acid as a product. The reaction equation is written as,
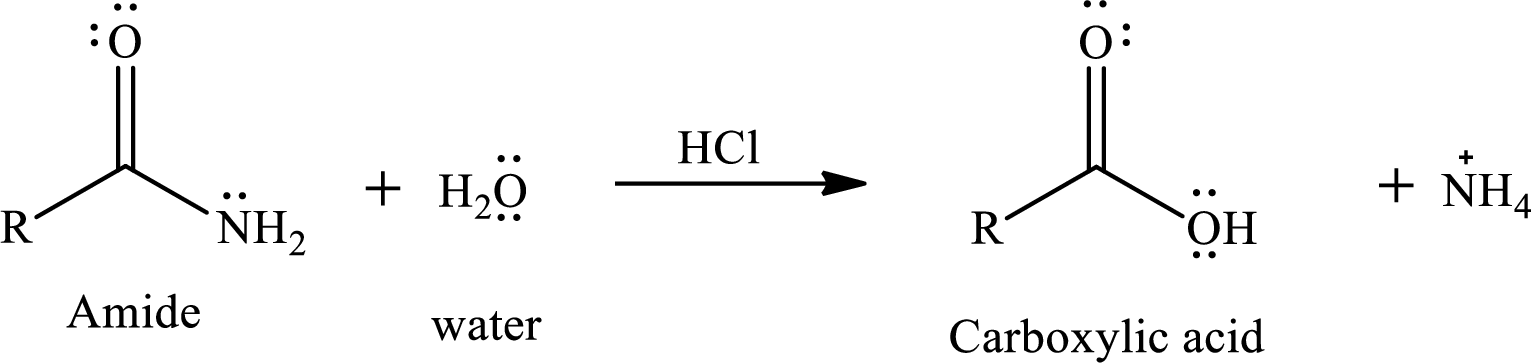
The reactivity of amide also depends upon the leaving tendency of amine part from acyl group. The weaker the base better will be the leaving group. The basicity of
(b)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
Amide hydrolysis in aqueous base:
A carboxylate salt and ammonia or an amine will be the product of amide hydrolysis in aqueous base.
For each mole of amide, one mole of base is required.
(c)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
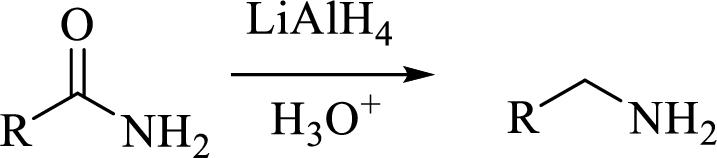
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry
- Draw a structural formula of the principal product formed when benzonitrile is treated with reagent. Q.) LiAlH4, then H2Oarrow_forwardDraw a structural formula of the principal product formed when benzonitrile is treated with reagent. Q.) H2O (excess), H2SO4, heatarrow_forwardComplete each of the following by supplying the missing reagents. Draw the structures of each of the reactants and products. a. N-Methylpropanamide + ? --> propanoic acid + ? b. N,N-Dimethylacetamide + strong acid --> ? + ? c. Formamide + strong acid --> ? + ?arrow_forward
- Draw a structural formula for the product of the reaction of acetophenone with reagent Q. HOCH2CH2OH, HClarrow_forwardDraw the condense structure for the following carboxylic acids: a) 2-propyamine b) 2-methyl-3-heptamine c) N-methylpentamine d) benzamide Write the reaction equation of hydrochloric acid, HCl, reacting with triethylamine (CH3CH2)3N. Write the reaction equation of benzamide reacting with sodium hydroxide (NaOH). write the full reactions for, the HCl hydrolysis of benzamide and acetamide and NaOH hydrolysis of benzamide and acetamide. From the equation for both Acid hydrolysis and Saponification, which reaction changes litmus paper from red to blue and whyarrow_forwardGive the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)NaHCO3, H2Oarrow_forward
- Amide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forwardWhich one of the following reacts with ethanoic acid and with phenol? A) Aqueous potassium hydroxide B) Bromine C) Calcium Carbonate D) Methanol and an acid catalystarrow_forwardIn addition to HCl, what is the product of the reaction of aprimary amine with an acid chloride? Draw the structureof that product and describe its featuresarrow_forward
- draw the structure of sodium fluoroacetate (Compound 1080, a controversial coyote poison)arrow_forwardGive the products formed when Benzaldehyde and Benzoic Acid are treated with the given reagents. a. Tollen’s reagentb. phenylhydrazine, H+c. HCNarrow_forwardGive the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)NaOH, H2Oarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




