
To describe: the bond holding the atoms together in this molecule.
Explanation of Solution
Introduction:
Bonds can be two types such as ionic bonding and covalent bonding. In ionic bond, transfer of electrons occurs and in case of covalent bond, sharing of electrons occurs.
The given diagram is shown below:
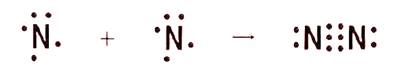
Nitrogen belongs to the group 15 and is a non-metal. The figure shows two nitrogen atoms. There are 5 valence electrons present on each nitrogen atom as shown in the figure.
The maximum number of electrons with which nitrogen can accommodate in its outermost shell to achieve stability is 8. Each nitrogen shares 3 electrons with other nitrogen to complete its outermost shell. Hence, this results in the triple bond between two nitrogen atoms.
The bond formed by the sharing of electrons between two nitrogen atoms is covalent bond. There is no charge separation between the nitrogen atoms. Thus, bond holding the nitrogen atoms together is also called a non-polar covalent bond.
Conclusion:
Hence, non-polar covalent bond holds the atoms together in this molecule.
Chapter 18 Solutions
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Additional Science Textbook Solutions
An Introduction to Thermal Physics
The Cosmic Perspective (8th Edition)
Introduction to Electrodynamics
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
University Physics (14th Edition)
Essential University Physics (3rd Edition)
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





