
Concept explainers
Interpretation:
Three possible structures for the constitutional isomers of molecular formula
Concept Introduction:
There are two main classes of isomers − constitutional isomers and stereo isomers.
Constitutional Isomers have the same molecular formula but differ in the way the atoms are connected to each other.
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
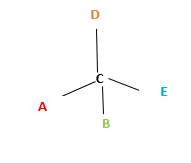
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different
Amine − is an organic N compounds formed by replacing one or more H atoms of
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw a 3-D structure of a molecule with a molecular formula of C4H9Cl containing one chiral center and draw its mirror image.arrow_forwardwhich is false? Enantiomers of amines are separable (resolvable). Amines are chiral. Shape of amines is trigonal pyramid. N in amines is sp3 hybridized.arrow_forwardDraw the structure of a compound of molecular formula C4H11NO that ts each description: (a) a compound that contains a 1° amine and a 3° alcohol; (b) a compound that contains a 3° amine and a 1° alcohol.arrow_forward
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forwarda. Classify each of the following amines as (1) primary, secondary or tertiary and as (2) aliphatic or aromatic. b. Classify each of the following amines as primary, secondary or tertiary.arrow_forwardEphedrine (2‑methylamino‑1‑phenylpropanol) is a drug that is used as a decongestant and bronchodilator to treat, among other conditions, the congestion due to the common cold. Place an asterisk (*) next to the chirality center(s) in the ephedrine molecule.arrow_forward
- Determine the relationship between the two compounds: A. Same compound B. Enantiomers C. Diastereomers D. Constitutional isomersarrow_forwardDraw molecules for the formula C5H10O with functional group alcohol and an ether with an R chiral center.arrow_forwardDraw and name compounds that meet the following descriptions a.Three acid chlorides having the formula C6H9ClO b. Three amides having the formula C7H11NOarrow_forward
- Determine if 2-methylocta-4,6-dien-1-amine contains any chiral carbons. If there are chiral carbons, circle or highlight all of them. If it does not contain any chiral carbons explain why none of the carbons are chiral.arrow_forward2. What is produced when an amine reacts with water? A. A primary alcohol and ammonia B. An amide and a hydrogen (H+) ion C. An ammonium ion and a hydroxide (OH-) ion D. An amide and a hydroxide (OH-) ionarrow_forwardDraw the structural formula for a compound with the given molecular formula. Q.)A chiral quaternary ammonium salt, C6H16NClarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



