
Concept explainers
(a)
Interpretation:
The atoms of 2-phenylethylamine in the molecule of mescaline needs to be determined.
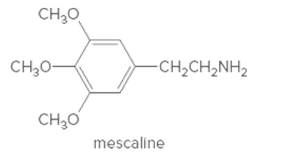
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In
(b)
Interpretation:
The atoms of 2-phenylethylamine in the molecule of LSD molecule needs to be determined.
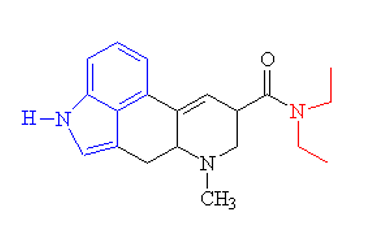
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Each H↔H eclipsing interaction in ethane costs about 4.0 kJ/mol. How many such interactions are percent in cyclopropane? What fraction of the overall 115 kJ/mol (27.5 kcal/mol) strain energy of cyclopropane is due to torsional strain.arrow_forwardGive a clear handwritten answer with explanation needed of each compound !!!arrow_forwarda. Explain why N-methylethanamine has a higher boiling point than trimethylamine even though they have the same molecular weight b. Explain the observed trend in the melting points for four isomers of molecular formula C7H16arrow_forward
- Finished A-C need help with the remaining D-Iarrow_forwardHelp with the following question Round the answer to 3 sig figsarrow_forwardExplain why hydrogen fluoride (Hf ) has a higher boiling temperature than hydrogen chloride ( HCL ) – 19.4 C vs 85 C – even though has a lower molecular weight.arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


