
(a)
Interpretation:
Whether the given molecule is a derivative of 2-phenylethylamine should be stated, if so, the 2-phenylethylamine unit within the molecule should be labeled.
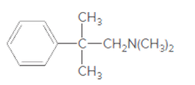
Concept Introduction:
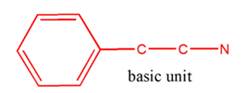
The general structure of 2-phenylethylamine is:
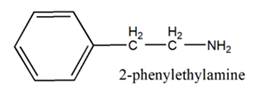
In 2-phenylethylamine, a benzene ring is bonded to a chain of two-carbon atoms that is bonded to a nitrogen atom.
So, the basic unit is of 2-phenylethylamine:
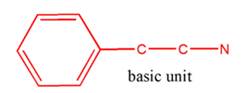
(b)
Interpretation:
Whether the given molecule is a derivative of 2-phenylethylamine should be stated, if so, the 2-phenylethylamine unit within the molecule should be labeled.
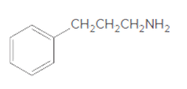
Concept Introduction:
The general structure of 2-phenylethylamine is:
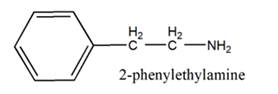
In 2-phenylethylamine, a benzene ring is bonded to a chain of two-carbon atoms that is bonded to a nitrogen atom.
So, the basic unit is of 2-phenylethylamine:
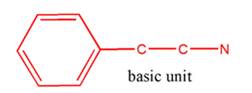
(c)
Interpretation:
Whether the given molecule is a derivative of 2-phenylethylamine should be stated, if so, the 2-phenylethylamine unit within the molecule should be labeled.
Concept Introduction:
The general structure of 2-phenylethylamine is:
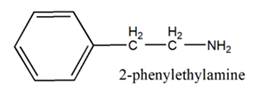
In 2-phenylethylamine, a benzene ring is bonded to a chain of two-carbon atoms that is bonded to a nitrogen atom.
So, the basic unit is of 2-phenylethylamine:
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Why is reductive amination a popular choice for the synthesis of amines? What are some other methods for the synthesis of amines?arrow_forwardA) Name the following amine. H3C−CH2−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound. B ) Name the following amine. CH3−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound.arrow_forwardWhich type of amine is (s)-methamphetamine? a) a primary aliphatic amine b) a primary aromatic amine c) a secondary aliphatic amine d) a secondary aromatic aminearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole





