
a)
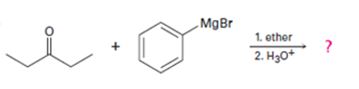
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
Ketones react with Grignard reagents to yield addition product which on acidification yield a tertiary alcohol a product.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
b)
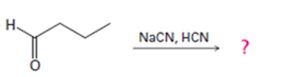
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
c)
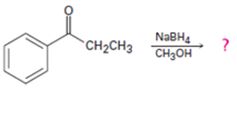
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
NaBH4 reduces aldehydes to primary alcohols and ketones to secondary alcohols. During the reaction the nucleophilic addition of hydrogen to the carbonyl group takes place.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
d)
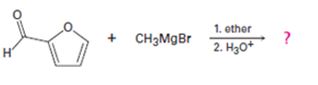
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
Aldehydes react with Grignard reagents to yield a secondary alcohol and with ketones to yield tertiary alcohols.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
Trending nowThis is a popular solution!

Chapter 19 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardProvide the product and mechanism for the reaction: ОН Cro, H,SO, H,0arrow_forward
- Predict the product(s) for each elimination reaction below. In each case show the mechanism. What do the mechanisms have in common? Why? (b) (c) CI CH3 CH3ONa CH3OH Br (CH3)3COK THE x OTos CH3ONa CH³OHarrow_forward9) Propose a mechanism that accounts for the following reaction. H2SO4 он 10) Predict the product or products for the following reaction. Suggest a mechanism for this reaction. Write structures for all product(s) of the steps. Circle the major product(s). heat + HBrarrow_forward+ hv H.2 + Propose a mechanism for the reaction that fully accounts for the formation of both products.arrow_forward
- Provide the major products for the following reactions?arrow_forward5. Provide the products necessary to complete the synthesis below. Indicate what mechanism was used for each product. Br H₂O H₂SO4 A B₂arrow_forwardGive the product(s) for the following reaction, predicting the major product, anc indicating by which mechanism each is formed. Br MeOH heatarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


