
a)
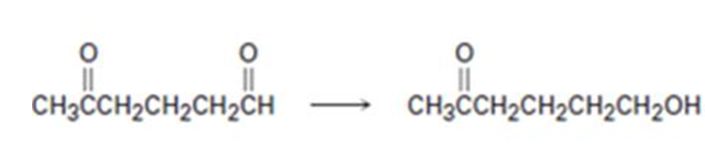
Interpretation:
How to carry out the selective transformation of a ketoaldehyde to ketoalcohol is to be shown.
Concept introduction:
Both
To show:
How to carry out the selective transformation of a ketoaldehyde to ketoalcohol.
b)
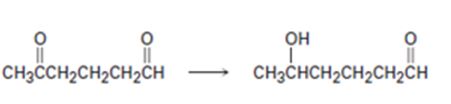
Interpretation:
How to carry out the selective transformation of a diketone to an aldol is to be shown.
Concept introduction:
Both aldehyde and keto groups are reduced by LiAlH4. In order to selectively reduce one keto group, the other keto group is first protected by converting it into an acetal. The protected
To show:
How to carry out the selective transformation of a diketone to an aldol.
Trending nowThis is a popular solution!

Chapter 19 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- H₂C ཏཱཏི 1 ནི OH 1. Br2, PBг3 2. H₂O H3C OH Br The a-bromination of carbonyl compounds by Br2 in acetic acid is limited to aldehydes and ketones because acids, esters, and amides don't enolize to a sufficient extent. Carboxylic acids, however, can be a-brominated by first converting the carboxylic acid to an acid bromide by treatment with PBr3. Following enolization of the acid bromide, Br2 reacts in an a-substitution reaction. Hydrolysis of the acid bromide completes the reaction. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions :0: H3C Br Br + :::OH2 Br H₂O H3C Br заarrow_forwardProvide reaction mechanisms for the following transformationsarrow_forwardWhich reagent(s), if any, may be used to carry out the following reaction?arrow_forward
- How might you carry out the following selective transformations? (Note that a protection step may be required, and recall that aldehydes are more reactive than ketones toward nucleophilic addition.)arrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents. cyclohexanol and BF3arrow_forwardSynthesize the following compounds using cyclohexanol, or any organic reagent that has 4 or fewer carbons, and any common reagents A) но, B) онarrow_forward
- How does propane synthesized from propanone? Show its mechanism.arrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrro- lidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place. Me Me Но N: 'N' AcO Ме pyrrolidinium acetate Me Me Mearrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forward
- 2 H3C H3C H C→XT OH H3C The aldol reaction is a carbonyl condensation reaction between two carbonyl partners and involves a combination of nucleophilic addition and a-substitution steps. One partner is converted into an enolate ion nucleophile and adds to the electrophilic carbonyl group of the second partner. In the classic aldol reaction, the carbonyl partners are aldehydes or ketones, although aldehydes are more reactive. The product is a ß-hydroxy carbonyl compound. base :0: OH H H Under reaction conditions slightly more vigorous than those employed for the aldol reaction, the ß-hydroxyl group is eliminated in an E1cB dehydration to give an a,ß-unsaturated carbonyl compound. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instruct ns H3C heat OH H3C :0: H + H₂O Harrow_forwardChoose reagents to convert 2-cyclohexenone to the following compounds.arrow_forwardThe reaction of an alkene with diazomethane forms a cyclopropane ring. Propose a mechanism for the reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

