
a)
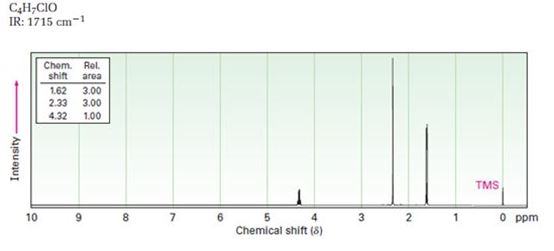
Interpretation:
A structure for ketone or aldehyde with the following descriptions is to be proposed.
C4H7ClO, IR: 1715 cm-1; 1HNMR: 1.62 δ (Rel.area=3.00), 2.33 δ (Rel.area=3.00), 4.32δ (Rel.area=1.00).
Concept introduction:
In 1HNMR the aldehyde protons absorb near 10 δ with a coupling constant , J = 3Hz. Hydrogens on the carbon next to aldehyde group absorb near 2.0-2.3 δ. Methyl ketones show a sharp three proton singlet near 2.1 δ.
To purpose:
A structure for ketone or aldehyde with the following descriptions.
C4H7ClO, IR: 1715 cm-1; 1HNMR: 1.62 δ (Rel.area=3.00), 2.33 δ (Rel.area=3.00), 4.32δ (Rel.area=1.00).
b)
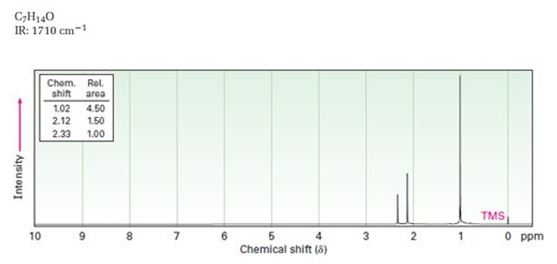
Interpretation:
A structure for ketone or aldehyde with the following descriptions is to be proposed.
C7H14O, IR: 1710 cm-1; 1HNMR: 1.02 δ (Rel.area=4.50), 2.12 δ (Rel.area=1.50), 2.33δ (Rel.area=1.00).
Concept introduction:
Aldehydes and ketones show a strong absorption band in IR from 1660-1770 cm-1. Aldehydes show two characteristic C-H absorptions between 2700-2760 cm-1 and 2800-2860 cm-1. Saturated aldehydes absorb near 1730 cm-1 while aromatic aldehydes and α, β- unsaturated aldehydes absorb near 1705 cm-1. Saturated ketones and cyclohexanones absorb near 1715 cm-1 while aromatic ketones and α, β- unsaturated ketones absorb near 1685-1690 cm-1. Cyclopentanones absorb around 1750 cm-1.
In 1HNMR the aldehyde protons absorb near 10 δ with a coupling constant , J = 3Hz. Hydrogens on the carbon next to aldehyde group absorb near 2.0-2.3 δ. Methyl ketones show a sharp three proton singlet near 2.1 δ.
To purpose:
A structure for ketone or aldehyde with the following descriptions.
C7H14O, IR: 1710 cm-1; 1HNMR: 1.02 δ (Rel.area=4.50), 2.12 δ (Rel.area=1.50), 2.33δ (Rel.area=1.00).
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- The 1H and 13C NMR spectra below belong to a compound with formula C6H10O2. Propose a structure for this compound.arrow_forwardPropose structures for alcohols that have the following 1HNMR spectra: (a) C5H12Oarrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forward
- Propose a structure for the compound, C4H8O, that has the following 1H NMR spectrumarrow_forwardAcid-catalyzed dehydration of 3-methyl-2-butanol gives three alkenes: 2-methyl-2- butene, 3-methyl-1-butene, and 2-methyl-1-butene. Propose a mechanism to account for the formation of each productarrow_forwardPhenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in over-thecounter pain relievers such as APC (aspirin, phenacetin, caffeine), phenacetin is no longer used because of its liver toxicity. Deduce the structure of phenacetin from its 1H NMR and IR spectra.arrow_forward
- Phenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in over-the-counter pain relievers such as APC (aspirin, phenacetin, caffeine), phenacetin is no longer used because of its liver toxicity. Deduce the structure of phenacetin from its 1H NMR and IR spectra.arrow_forwardWhat is the correct assignment of the names of the following ketones? H;C CH3 CH3 1 2 O 1 = acetone; 2 = phenol; 3 = benzaldehyde 1 = acetone; 2 = acetophenone; 3 = benzophenone %3D %3D O 1 = formaldehyde; 2 = benzaldehyde; 3 = acetophenone %3D O 1 = acetaldehyde; 2 = acetophenone; 3 = benzaldehyde %3D %3Darrow_forwardProtons on a carbon atom located adjacent to the carbonyl group generally absorb in the range of 1-2.5 ppm. Protons on a carbon atom located adjacent to an oxygen atom generally absorb in the range of 3.5-5 ppm. What is the structure of the ester from the Proton NMR spectrum?arrow_forward
- Propose the structure of a compound that exhibits the following ¹H NMR data. C5H10O 1.09 ppm (6H, doublet) 2.12 ppm (3H, singlet) 2.58 ppm (1H, septet)arrow_forwardPropose a mechanism for the acid-catalyzed hydration of methylidenecyclohexane to give 1-methylcyclohexanol. Which step in your mechanism is rate-determining?arrow_forwardPropose a mechanism for the acid-catalyzed hydration of 1-methylcyclohexene to give 1-methylcyclohexanol. Which step in your mechanism is rate-determining?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


