
The pH of human stomach acid ranges from 1.0 to 3.0, whereas a healthy esophagus has a pH of approximately 7.0. In gastroesophageal reflux disease (GERD), often called acid reflux, stomach acid flows backward from the stomach into the esophagus. Repeated episodes in which esophageal pH goes below 4.0, considered clinical acid reflux, can result in bleeding ulcers and damage to the esophageal lining. The data in the Figure, from a patient with GERD, show esophageal pH during a sleeping reflux event.
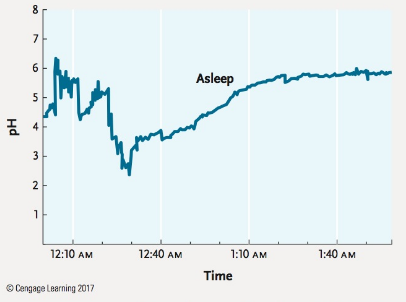
How many minutes does it take to go from the peak of the reflux event (when pH is most acidic) to when the reflux event is over?
Source: Based on T. Demeester et al. 1976. Patterns of gastroesophageal reflux in health and disease. Annals of Surgery 184:459–469.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Biology: The Dynamic Science (MindTap Course List)
- Carbohydrates and lipids are composed of the same chemical element, but in different proportions. Both are used primarily as energy source for metabolic process. Which type of biomolecule has the higher calorie content per gram? Explain briefly.arrow_forwardIndicate whether each of the following is due to an increase in pH, a decrease in pH or neither and explain why: __Makes carrots soften more when cooked __Color shifts due to adding chlorine to cake flour __Lowers the amount of iron sulfide formed in hard cooked eggs __Makes anthocyanins more red __Makes an egg white foam more stable __Makes starch dextrinize when heated in boiling water __Raises osmotic pressurearrow_forwardIn the course of a hypothetical meal, the stomach expands from 0.25 liters to 1.25 liters. If we assume that no buffering occurs and no acid is produced from the stomach contents, how much acid must be secreted to maintain pH = 1.0? How many grams of ATP would be consumed in the production of that acid?arrow_forward
- What effect on total protein concentration would you expect to see during dehydration? Explain your reasoning.arrow_forwardcalculate the amount of that food that you would need to ingest for it to be fatal to a human assuming there is a 5 l of blood in the human body is this an amount that you need to be concerned about? explain amount needed: 5ml Final pH: 5.8arrow_forwardA patient was rushed to the hospital with the following signs and symptoms. Tingling sensation of the hands and feet, polydipsia, polyuria, blurred vision & unexplained weight loss. The lab results revealed the following: pH = 7.45 HCO3 = 28 mmol/L and pCO2 = 27mmHg. What will be the compensation of the body? Provide comprehensive explanations to justify your answer.arrow_forward
- Drugs are small molecules that often occur in either a neutral or a charged form, depending on pH. For example, aspirin can be either neutral or lose a proton to be negatively charged. When aspirin is charged, it does not diffuse well into the body. pH varies in the gastrointestinal track. Since the stomach has a low pH (pH=3), it has a larger concentration of neutral aspirin (10-3 M), while the small intestine (pH=6) has a smaller neutral aspirin concentration (10-6 M). For the following, assume that the neutrally charged aspirin molecule has a diffusion coefficient through the cell of 1 x 10-6 cm2/s. C) We’d like to figure out where in the body aspirin is absorbed as determined by its diffusion rate. If the small intestine has a surface area 50 times that of the stomach, what is the ratio of the diffusion rates in the stomach compared to the small intestine? D) In which part of the GI tract is most of the aspirin absorbed?arrow_forwardConsider the scenario below: A 40-year-old woman was rushing to get to her workplace before office time. What do you think would have happened to her blood's pH? _________________________________________________________________ _________________________________________________________________ What would be the effect on pH once she reaches her office, but continues to breathe hard for a few minutes? __________________________________________________________________ __________________________________________________________________ Why? __________________________________________________________________ __________________________________________________________________ A weak acid causes her issue. She ingested far too much aspirin, also known as acetylsalicylic acid, and ended up in the emergency room (ER). The pH range within which humans can survive is between pH 6.8 and about pH 8.0. The woman's pH is outside of this window. What do you think would have happened to her?…arrow_forwardIndividuals suffering from ketoacidosis have acidic urine. What effect do you expect ketones to have on pH? Why is pH lowered when ketone bodies are present?arrow_forward
- The pH scale shows availability of reactive hydrogen ions (H+) in a liquid. The scale is logarithmic, so milk has 10 times as many H+ ions as pure water, for a given volume. How many more H+ ions does soda pop have compared to pure water? Write the number only.arrow_forwardProteasas are enzymes that break down proteins. Gastric proteasas are found in the stomach; intestinal proteases are found in the small intestine. Estimate the optimum pH for these two enzymes and identify as acidic or basic: Gastric proteases: pH= ____; acidic or basic Intestinal proteases activity: pH= ____; acidic or basicarrow_forwardBased on the pH of your drug, if your drug was in a high concentration in the blood, how would it affect the pH of your blood. Would it cause acidosis or alkalosis, and how would you treat this? the drug name is captopril and the pH is 2.65arrow_forward
 Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage LearningNutritional Sciences: From Fundamentals to Food, ...Health & NutritionISBN:9781337486415Author:McGuirePublisher:Cengage
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage LearningNutritional Sciences: From Fundamentals to Food, ...Health & NutritionISBN:9781337486415Author:McGuirePublisher:Cengage BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning



