
Concept explainers
(a)
Interpretation:
The electron dot structure of Beryllium has to be given.
Concept Introduction:
Electron dot structure:
A Lewis dot diagram is a representation of the valence electrons of an atom that uses dots around the
Example:
The valence electron of Mg is two it can be represented in electron dot structure as,
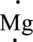
(b)
Interpretation:
The electron dot structure of Iodine has to be given.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The electron dot structure of Magnesium has to be given.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The electron dot structure of Argon has to be given.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- What is the maximum number of electrons in an n = 2 shell?arrow_forwardWhat would be your reaction to a claim that a new element had been discovered, and it fit between tin (Sn) and antimony (Sb) in the periodic table?arrow_forward2. Give the 4 sets of quantum numbers for the 16 electrons of Sulfur.arrow_forward
- Write the complete ground-state electron configuration of S²⁻.arrow_forwardThe ionic radii of the ions S2–, Cl–, and K+ are 184, 181, 138 pm respectively. Explain why these ions have different sizes even though they contain the same number of electrons.arrow_forwardExplain the organization of an atom of carbon.arrow_forward
- Give the symbol of the element that has the largest atomic size in Period 1.arrow_forwardConsider the following elements – O, F, Na, Mg and Al. -What is the first criterion you would use to determine which the largest atom? Explain why you chose this as the first criterion.-Are there any other criteria you would use to determine the next largest atom?-Arrange the above atoms in ascending order (smallest to largest). Explain your reasoningarrow_forward2.84 Early attempts to arrange the elements often focused on atomic weight. Mendeleev considered a number of properties in addition to atomic weight, so he realized that some elements seemed out of place when ordered by atomic weight. Using the modern periodic table, identify elements for which Mendeleev must have had to switch the order in order to get the correct sequence of elements.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





