
Concept explainers
(a)
Interpretation:
The species with highest mass number among the given figure has to be given.
The given figure is,
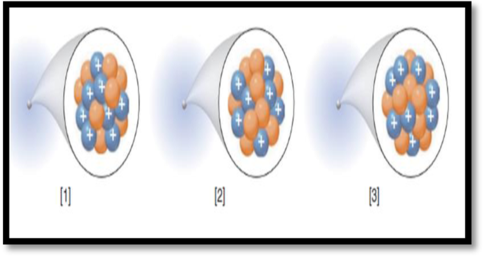
Figure 1
Concept Introduction:
Mass number (A):
Mass number of an element is defined as the total number of protons and neutrons in an atom.
(b)
Interpretation:
The number of electrons in each species given in the representation has to be given.
The given figure is,
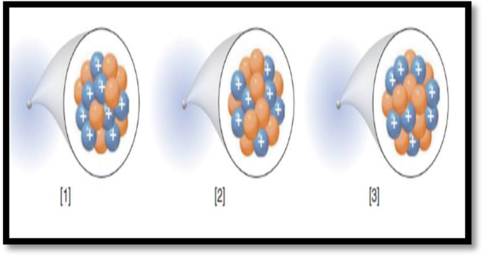
Figure 1
Concept Introduction:
Electrons:
Electrons have an electric charge of
(c)
Interpretation:
The isotopic symbol of each species in the given figure has to be given.
The given figure is,
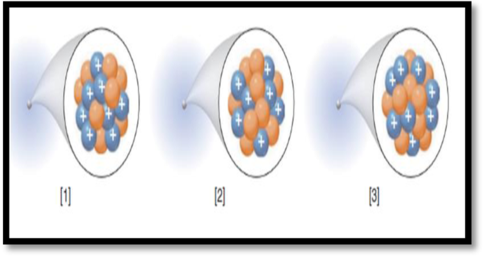
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- Is it possible for the same Iwo elements to form more than one compound? Is this consistent with Dalton’s atomic theory? Give an example.arrow_forwardAverage atomic masses listed by JUPAC are based on a study of experimental results. Bromine has two isotopes 79Br and 81Br, whose masses (78.9 183 and 80.9 163 amu) and abundances (50.69% and 49.3 1%) were determined in earlier experiments. Calculate the average atomic mass of bromine based on these experiments.arrow_forwardA cube of sodium has length 1.25 in. How many atoms are in that cube? (Note: dNa=0.968g/cm3.)arrow_forward
- Write symbols like those given in Exercises 2.15 and 2.16 for the following isotopes: a. silicon -28 b. argon -40 c. strontium -88arrow_forwardAnswer the questions below, using LT (for is less than), GT (for is greater than), EQ (for is equal to), or MI (for more information required) in the blanks provided. (a) The mass (to three significant figures) of6.0221023atoms of Na 23.0 g. (b) Boron has two isotopes, B-10 (10.01 amu) and B-11 (11.01 amu). The abundance of B-10 the abundance of B-11. (c) If S-32 were assigned as the standard for expressing relative atomic masses and assigned an atomic mass of 10.00 amu, the atomic mass for H would be 1.00 amu. (d) When phosphine gas, PH3, is burned in oxygen, tetraphosphorus decaoxide and steam are formed. In the balanced equation (using smallest whole-number coefficients) for the reaction, the sum of the coefficients on the reactant side is 7. (e) The mass (in grams) of one mole of bromine molecules is 79.90.arrow_forwardWhy is it important to understand atoms?arrow_forward
- Calculate the precise mass of each ion to five significant figures. Unless otherwise indicated, use the mass of the most abundant isotope of each element. Q.)[13CH3Br]+arrow_forwardSycamorium Sy, a hypothetical element occurs in three isotopes, who masses and abundances are given below Isotope1 has a mass of 297.021 with an abundance of 21.9 %; Isotope 2 has a mass of 299.010 withan abundance of 33.3 %; the rest is isotope 3 with a mass of 301.057. Calculate the atomic mass ofsycamoriumarrow_forwardWhen writing a complete atomic symbol, there are four numbers used – in the upper-left, upper-right, lower-left, andlower-right corners. (i) State what each of these numbers represents. Then, (ii) state which of the numbers differ(s)between isotopes and (iii) explain the reasoning behind your choicearrow_forward

 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





