
(a)
Interpretation:
The atom with smaller atomic radius has to be given.
The given diagram is,
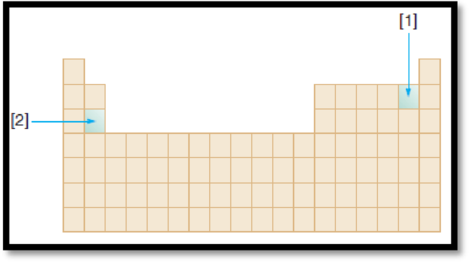
Figure 1
Concept Introduction:
Atomic Radius:
Atomic radius is the distance from the centre of the nucleus to the outermost shell containing electrons. The atomic radius of an element decreases along the period and increases down the group.
(b)
Interpretation:
The atom with lower ionization energy has to be given.
The given diagram is,
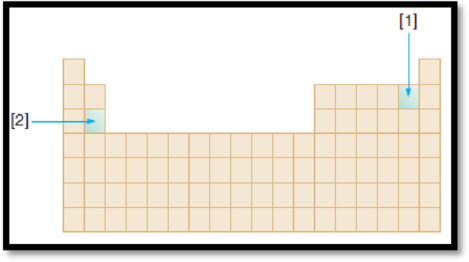
Figure 1
Concept Introduction:
Ionization energy:
Ionization energy is defined as the energy required removing one mole of electron from one mole of isolated gaseous atom or ion. Ionization energy of an element increases along the period and decreases down the group.
(c)
Interpretation:
The elements in the diagram have to be identified.
The given diagram is,
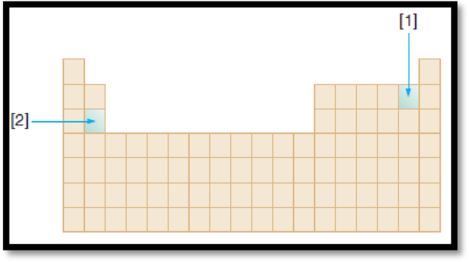
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





