
Concept explainers
(a)
Interpretation:
The number of protons and the number of neutrons for the atom in the given figure has to be given.
The given figure is,
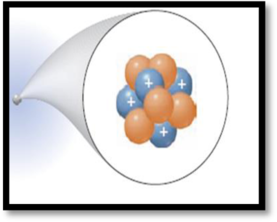
Figure 1
Concept Introduction:
Protons:
A proton is one of three main particles that make up the atom. Protons are a tiny, dense region at the center of the atom. Protons have a positive electrical charge of
Neutrons:
Atoms of all elements except for most atoms of hydrogen have neutrons in their nucleus. Unlike protons and electrons, which are electrically charged, neutrons have no charge they are electrically neutral.
(b)
Interpretation:
The
The given figure is,
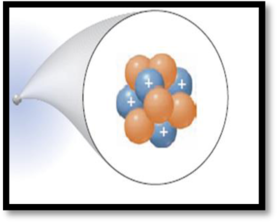
Figure 1
Concept introduction;
Atomic number (Z):
Atomic number of an element is the number of protons found in the nucleus of every atom of that element.
Mass number (A):
Mass number of an element is defined as the total number of protons and neutrons in an atom.
(c)
Interpretation:
The element for the given atom has to be identified.
The given figure is,
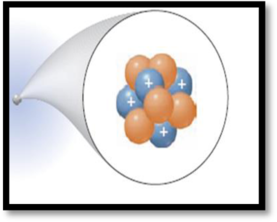
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- Define the term atomic weight. Why might the values of atomic weights on a planet elsewhere in the universe be different from those on earth?arrow_forwardThe following table presents the abundances and masses of the isotopes of zinc. What is the atomic weight of zinc?arrow_forwardHow is an atom’s atomic number related to its number ofprotons? To its number of electrons?arrow_forward
- What are the different kinds of particles in the atoms nucleus? Compare their properties with each other and with those of an electron.arrow_forwardThough the common isotope of aluminum has a mass number of 27, isotopes of aluminum have been isolated (or prepared in nuclear reactors) with mass numbers of 24, 25, 26, 28, 29, and 30. How many neutrons are present in each of these isotopes? Why are they all considered aluminum atoms, even though they differ greatly in mass? Write the atomic symbol for each isotope.arrow_forwardWhat advantage does the atomic mass unit have over grams when speaking of the mass of an atom or a subatomic particle?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





