
(a)
Interpretation:
The atom has higher
The given diagram is,
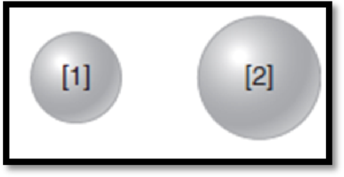
Figure 1
Concept Introduction:
Atomic number (Z):
Atomic number of an element is the number of protons found in the nucleus of every atom of that element.
(b)
Interpretation:
The atom has higher ionization energy has to be given.
The given diagram is,
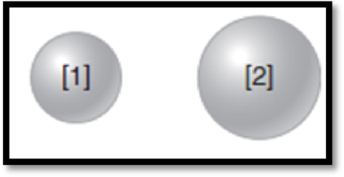
Figure 1
Concept Introduction:
Ionization energy:
Ionization energy is defined as the energy required removing one mole of electron from one mole of isolated gaseous atom or ion.
Periodic trends:
Ionization energy of an element increases along the period and decreases down the group.
(c)
Interpretation:
The atom has higher ionization energy has to be given.
The given diagram is,
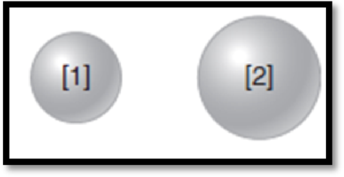
Figure 1
Concept Introduction:
Refer to part (b).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- Is there any change in the radius of the atom when it transforms into an ion?arrow_forwardOf the elements Be, C, Cl, Sb, and Cs, which is most likely to be a metalloid?arrow_forwardIn the following set of elements, rank them from LOWEST first ionization energy to HIGHEST first ionization energy. He, Ar, Mg, Al, P, Clarrow_forward
- Would you expect an anion to be larger or smaller than the corresponding atom, and why?arrow_forwardClassify each element in the fourth row of the periodic table as a metal, nonmetal, or metalloid.arrow_forward(a) What is the general relationship between the size of anatom and its first ionization energy? (b) Which element inthe periodic table has the largest ionization energy? Whichhas the smallest?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





