
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 38P
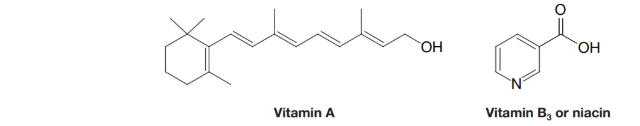
(a) Indicate the hydrophobic and hydrophilic parts of vitamin A and comment on whether you would expect it to be soluble in water. (b) Do the same for vitamin
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rationalize the fact that hydrogen bondinghas not been observed between CH4 molecules.
Question:
What is the impact of molecular chirality on the pharmacological activity of drugs, and how can it be harnessed for the development of more effective therapeutic agents?
Describe the layers that can form in a separatory funnel in extracting caffeine from tea leaves and the general properties of its contents.
Chapter 2 Solutions
Organic Chemistry
Ch. 2 - Practice Problem 2.1
Propose structures for two...Ch. 2 - Prob. 2PPCh. 2 - Prob. 3PPCh. 2 - Prob. 4PPCh. 2 - Prob. 5PPCh. 2 - Practice Problem 2.6
Using a three-dimensional...Ch. 2 - Practice Problem 2.7
Trichloromethane (, also...Ch. 2 - Prob. 8PPCh. 2 - Prob. 9PPCh. 2 - Practice Problem 2.10
Write bond-line structural...
Ch. 2 - Practice Problem 2.11 Although we shall discuss...Ch. 2 - Practice Problem 2.12 Write bond-line structural...Ch. 2 - Prob. 13PPCh. 2 - Practice Problem 2.14
One way of naming ethers is...Ch. 2 - Practice Problem 2.15 Eugenol is the main...Ch. 2 - Practice Problem 2.16
One way of naming amines is...Ch. 2 - Practice Problem 2.17 Which amines in Practice...Ch. 2 - Prob. 18PPCh. 2 - Prob. 19PPCh. 2 - Practice Problem 2.20
Write bond-line formulas for...Ch. 2 - Practice Problem 2.21
Write bond-line formulas for...Ch. 2 - Practice Problem 2.22
Write bond-line formulas for...Ch. 2 - Prob. 23PPCh. 2 - Practice Problem 2.24 Write another resonance...Ch. 2 - Prob. 25PPCh. 2 - Practice Problem 2.26
Which compound would you...Ch. 2 - Practice Problem 2.27 Arrange the following...Ch. 2 - Prob. 28PPCh. 2 - Prob. 29PCh. 2 - Identify all of the functional groups in each of...Ch. 2 - 2.31 There are four alkyl bromides with the...Ch. 2 - Prob. 32PCh. 2 - Classify the following alcohols as primary,...Ch. 2 - 2.34 Classify the following amines as primary,...Ch. 2 - Prob. 35PCh. 2 - Identify all of the functional groups in Crixivan,...Ch. 2 - 2.37 Identify all of the functional groups in...Ch. 2 - 2.38 (a) Indicate the hydrophobic and hydrophilic...Ch. 2 - Hydrogen fluoride has a dipole moment of 1.83 D;...Ch. 2 - 2.40 Why does one expect the cis isomer of an...Ch. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - 2.44 Consider each of the following molecules in...Ch. 2 - True or false: For a molecule to be polar, the...Ch. 2 - 2.46 Which compound in each of the following...Ch. 2 - Prob. 47PCh. 2 - The IR spectrum of propanoic acid (Fig. 2.16)...Ch. 2 - Prob. 49PCh. 2 - Write structural formulas for four compounds with...Ch. 2 - There are four amides with the formula C3H7NO. (a)...Ch. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - 2.56 Compound C is asymmetric, has molecular...Ch. 2 - 2.57 Examine the diagram showing an -helical...Ch. 2 - Prob. 1LGPCh. 2 - Prob. 2LGPCh. 2 - Prob. 3LGPCh. 2 - Consider the molecular formula C4H8O2. Predict...Ch. 2 - Consider the molecular formula C4H8O2. If any of...Ch. 2 - Prob. 6LGPCh. 2 - Consider the molecular formula.
7. Pick five...Ch. 2 - Prob. 8LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
A liquid mixture contains 60.0 wt% ethanol (E), 5.0 wt% of a dissolved solute (S), and the balance water. A str...
Elementary Principles of Chemical Processes, Binder Ready Version
Q4. Which property of rubbing alcohol is a chemical property?
a) Density (0.786 g/cm3)
b) Flammability
c) Bo...
Chemistry: A Molecular Approach (4th Edition)
For each of the following molecules, (1) draw the correct Lewis structure; (2) label each polar covalent bond w...
General, Organic, and Biological Chemistry (3rd Edition)
9.1 Calculate the total mass of the reactants and the products for each of the following equations:
Basic Chemistry (5th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
1.2 Ask two of your friends (not in class) to define the terms in problem1.1.
Do their answers agee with the d...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain why cholesterol, a compound with molecular formula C 27H 46O and one OH group, is soluble in CCl 4 but insoluble in water.arrow_forwardA chemist, during the course of an analysis, runs across a chemical composed of carbon, hydrogen, and oxygen in the proportion 1:2:1 and having a six-sided molecular shape. It is probably (a) a pentose, (b) an amino acid, (c) a fatty acid, (d) a monosaccharide, (e) a nucleic acid.arrow_forwardWhich of the following in each pair is likely to be moresoluble in hexane, C6H14: (a) CCl4 or CaCl2, (b) benzene(C6H6) or glycerol, CH2(OH)CH(OH)CH2OH, (c) octanoicacid, CH3CH2CH2CH2CH2CH2CH2COOH, or acetic acid,CH3COOH? Explain your answer in each case.arrow_forward
- Some proteins reside in the hydrophobic lipid bilayer of the cell membrane. Would hydrophilic groups of these proteins still be facing the lipid “solvent”?arrow_forward(a) Draw four compounds with molecular formula C6H12O, each containing at least one different functional group, (b) Predict which compound has the highest boiling point, and explain your reasoning.arrow_forwardIntramolecular forces of attraction are often important in holding large molecules together. For example, some proteins fold into compact shapes, held together by attractive forces between nearby functional groups. A schematic of a folded protein is drawn here, with the protein backbone indicated by a blue-green ribbon, and various appendages drawn dangling from the chain. What types of intramolecular forces occur at each labeled site (A–F)?arrow_forward
- what intermolecular forces are present in quaternary ammonium salts? are there any chiral centers present in that substance? how do individual molecules in that class of pesticide interact with eachother?arrow_forwardIdentify the two layers in the procedure of caffeine extraction from tea bags. In which part does caffeine can be extracted?arrow_forwardDiscuss the role of the hydrophobic interactions in stabilizing the following. Q.) Lipid bilayersarrow_forward
- Determine the molecular geometry and polarity of hydroxylamine (NH2OH), formaldehyde (CH2O), and formaldoxime (H2CNOH). Justify your deductions. Is there a difference between reactants and products for both features?.arrow_forwardIntramolecular forces of attraction are often important in holding largemolecules together. For example, some proteins fold into compactshapes, held together by attractive forces between nearby functionalgroups. A schematic of a folded protein is drawn here, with the proteinbackbone indicated by a blue-green ribbon, and various appendagesdrawn dangling from the chain. What types of intramolecular forcesoccur at each labeled site (A–F)?arrow_forwardUsing your own words and illustrations, concisely discuss the absorption of triglycerides, phospholipids, and cholesterol esters from food. Give answer all questions with explanation.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY