
Practice Problem 2.1
Propose structures for two constitutional isomers of cyclopentene that do not contain a ring.
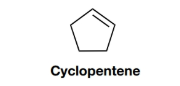
Interpretation:
The structuresfor two constitutional isomers of cyclopentene, which do not contain a ring, are to be proposed.
Concept introduction:
When two molecules have the same molecular formula, they are called isomers. If the connectivity of atoms is different, they are called constitutional isomers.
Constitutional isomers are the isomers in which the molecular formula remains the same but the arrangement of groups or atoms are different in structure.
In order to estimate the number of constitutional isomers of a particular compound, count the number of each atom and arranged all the atoms in different ways.
Answer to Problem 1PP
Solution:


Given: The structure of cyclopentene is as follows:
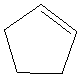
Explanation of Solution
The structure of cyclopentane contains 5 carbon atoms and 8 hydrogen atoms. Thus, the molecular formula of cyclopenteneis
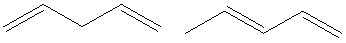
The molecular formula is
Hence, the two constitutional isomers of

Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry
Organic Chemistry (8th Edition)
Fundamentals of Heat and Mass Transfer
Chemistry & Chemical Reactivity
- Give the IUPAC name in polyfunctional compound.arrow_forwardGive typed full explanation not a single word hand written otherwise leave it Spell out the full name of each compound. A, B, C, & Darrow_forwardGive a clear handwritten answer with clearly....complete the given below formula...with their name and formatearrow_forward
- Fill the empty boxes with compatible structure. (Give answer in clear handwritten)arrow_forwardIn the space provided below, write a line structure corresponding to the systematic name (R)-3-methylhexane. Then, provide a Newman projection that illustrates the lowest energy conformation of (R)-3-methylhexane along the C3-C4 bond axis of the same. Again, place your answer in the space provided.arrow_forwardCompound D and compound E are in equilibrium. Write a stepwise mechanism from compound D to compound E showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show necessary lone pairs and all formal charges. Lastly explain which compound (D or E) will be in higher concentration.arrow_forward
- Give a clear handwritten answer with explanation needed..given below some options in which compound is trans-1,3-dimethylcyclohexane please give answer in detailedarrow_forwardDraw the structural formulas of compounds A, C, D, E and F and draw the isomer of B, with the explaination on which one would be the major product and why.arrow_forwardUsing the degrees of unsaturation, determine the molecular formula for the following compound (NOT use subscripts )arrow_forward
- Provide the least stable conformation of the following compound. Iarrow_forwardGive a clear handwritten answer with explanation..give the most stable chair conformation given bleow structures....please give answer all sub partsarrow_forwardDraw the Newmann projections of this molecule along the circled bond of this molecule, from the point of view of the eye. You should include and label the syn-periplanar, gauche, anticlinal and anti-periplanar conformations, and whether the conformation is staggered or eclipsed. List these four conformations from most stable to least stable?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





