
Concept explainers
(a)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
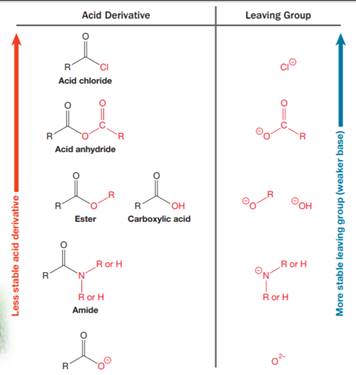
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
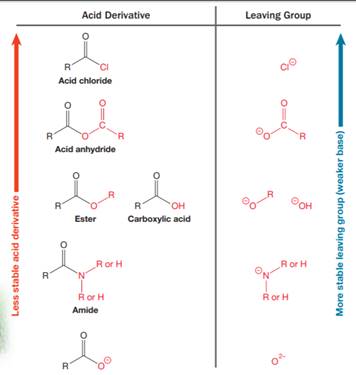
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and the reaction does not occur readily.
Explanation of Solution
The given reaction is
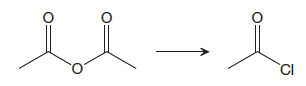
This is an example of a nucleophilic acyl substitution reaction in which an anhydride is getting converted into an acyl chloride. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
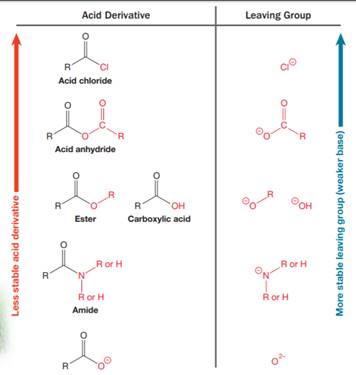
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to the one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an acid chloride is from a lower rung while an acyl chloride is from a higher rung on the stability ladder. Thus, this reaction is energetically unfavorable and so does not occur readily.
Conversion of an acid derivative from a lower rung to higher rung on the stability ladder is energetically unfavourable, and the reaction does not occur readily.
(b)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
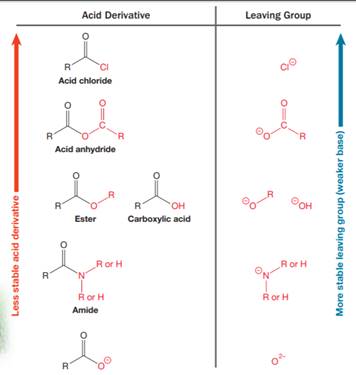
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and it does not occur readily.
Explanation of Solution
The given reaction is
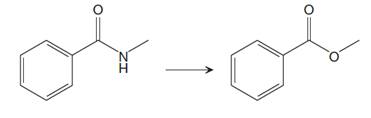
This is an example of a nucleophilic acyl substitution reaction in which an amide is getting converted into an ester. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
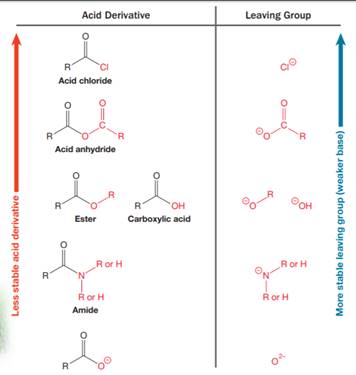
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an amide is from a lower rung while an ester is from a higher rung on the stability ladder. Thus, this reaction is energetically unfavorable and so does not occur readily.
Conversion of an acid derivative from a lower rung to higher rung on the stability ladder is energetically unfavourable, and the reaction does not occur readily.
(c)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
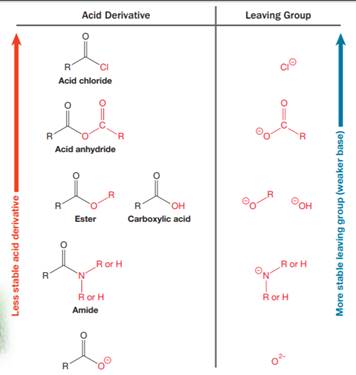
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and it does not occur readily.
Explanation of Solution
The given reaction is
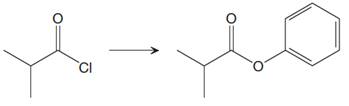
This is an example of nucleophilic acyl substitution reaction in which an acid chloride is getting converted into an ester. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an acid chloride is from a higher rung while an ester is from a lower rung on the stability ladder. Thus, this reaction is energetically favorable and occurs readily.
Conversion of an acid derivative from a higher rung to lower rung on the stability ladder is energetically favourable, and the reaction occurs readily.
(d)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:

An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and it does not occur readily.
Explanation of Solution
The given reaction is
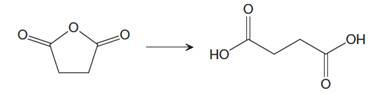
This is an example of a nucleophilic acyl substitution reaction in which an acid anhydride is getting converted into dicarboxylic acid. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:

An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an acid anhydride is from a higher rung while a carboxylic acid is from a lower rung on the stability ladder. Thus, this reaction is energetically favorable and occurs readily.
Conversion of an acid derivative from a higher rung to lower rung on the stability ladder is energetically favourable, and the reaction occurs readily.
Want to see more full solutions like this?
Chapter 20 Solutions
ORG.CHEM W/TEXT+SOLU.MANUAL
- Predict/draw the major product of the reaction shown in the picture:arrow_forward(SYN) In the reaction shown here, the aromatic ring has just one chemically distinct, aromatic H, so a single electrophilic aromatic substitution will lead to just a single product. With this in mind, supply the missing reagentsneeded to carry out the transformation.arrow_forwardIn (e), please note that the heterocyclic oxygen atoms contains two lone pairs that could contribute to the resonance stabilization of any proposed intermediate.arrow_forward
- Textbook problem: Suggest short series of reactions that would be expected to transform the material on the right into the desired product shown on left.arrow_forwardDirection: predict the major new product if any resulting from the each of the following reactions please use this example as example. Dont use the stick form those are confusing.arrow_forwardOn each of the reactions shown attached please indicate if the reaction will take place or not and if the reaction will not take place explain why also if the reaction will take place also explain why then draw the productarrow_forward
- Please solve.... what is the major product?with explanation needarrow_forwardWhich of the following would proceed with the fastest rate? Why? Options: top reaction due to stronger base bottom reaction due to stronger base top due to less hindered substrate bottom due to more hindered substratearrow_forwardPlease draw the major product/products of each of the following reactionarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
