
Concept explainers
Give the structure corresponding to each name.
a.
b.
c.
d.
e.
f.
g.
h.
(a)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
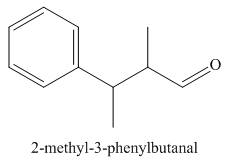
Figure 1
Explanation of Solution
The given name is
The structure corresponding to
(b)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
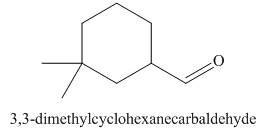
Figure 2
Explanation of Solution
The given name is
The structure corresponding to
(c)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
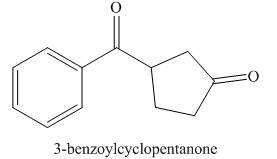
Figure 3
Explanation of Solution
The given name is
The structure corresponding to
(d)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
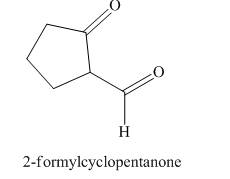
Figure 4
Explanation of Solution
The given name is
The structure corresponding to
(e)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
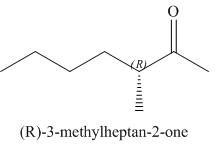
Figure 5
Explanation of Solution
The given name is
The structure corresponding to
(f)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
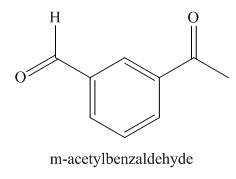
Figure 6
Explanation of Solution
The given name is
The structure corresponding to
(g)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
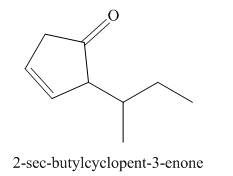
Figure 7
Explanation of Solution
The given name is
In the given compound, one benzaldehyde group is present. The functional group present in the given compound is ketone.
The structure corresponding to
(h)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ene.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 21.44P
The structure corresponding to
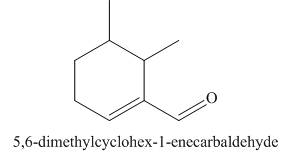
Figure 8
Explanation of Solution
The given name is
The structure corresponding to
Want to see more full solutions like this?
Chapter 21 Solutions
ORGANIC CHEMISTRY
- Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridinearrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forwardDraw the structure corresponding to each name. a. 5-methylheptanoyl chloride b. isopropyl propanoate c. acetic formic anhydride d. N-isobutyl-N-methylbutanamide e. 3-methylpentanenitrile f. 0-cyanobenzoic acid g. sec-butyl 2-methylhexanoate h. N-ethylhexanamidearrow_forward
- Instructions: Draw out each compound to clearly show what groups are bonded to the carbonyl carbon. Label each compound as a carboxylic acid, ester, or amide. a. CH3CH2CO2CH2CH3 b. CH3CONHCH3 c. (CH3)3CCO2H d. (CH3)2CHCON(CH3)2 Instructions: Give the IUPAC name for each compound. A. CH₂ CH₂CH₂CH₂CCH₂COOH CH3 B. CH₂CHCH₂CH₂COOH CH₂COOH CH₂CH3 C. (CH,CH,),CHCH,CHCOOH Instructions: Give the structure corresponding to each IUPAC name. a. 2-bromobutanoic acid b. 2,3-dimethylpentanoic acid c. 2-ethyl-5,5-dimethyloctanoic acid d. 3,4,5,6-tetraethyldecanoic acidarrow_forwardNonearrow_forwardstructure onlyarrow_forward
- Which is propyl propanoate? A. CH₂CH₂CH₂OOCCH₂CH; B. CH₂CH₂CH₂COOCH₂CH₂ C. CH₂CH₂CH₂COCH₂CH₂ D. CHỊCH,CH,OCH,CHỊCH, A B C Darrow_forwardThere are THREE parts. Please circle the final answer! Which reagent would not react with a carboxylic acid? O a. LIAIH4 b. NaOH C. KMN04 d. ВНЗ/THF An aldehyde cannot be made directly from this type of compound a. carboxylic acid O b. alcohol C. ester O d. acid chloridearrow_forwardWhat is an acceptable name for this compound? OH NO₂ 2-nitro-phentanol B. 1-hydoxy-2-nitrobenzene C. o-nitrophenol D. 1-nitrophenolarrow_forward
- Which of the compounds is/are hemiacetal, acetal, hemiketal and ketal?arrow_forward1. An alkene reacts with water with an acid catalyst results into a formation of: A. Aldehyde B. Ketone C. Alcohol D. Ester 2. 3-Methylhexanal with K2Cr2O7 will yield: A. 3-Methyl-1-hexanol B. 3-Methylhexanoic acid C. 3-Methyl-1-hexanone D. 3-Methyl-1-hexanethiol 3. This is a reverse process of Hydration reaction: A. Oxidation reaction B. Reduction reaction C. Dehydration reaction D. Hydration reaction 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forwardDraw the products of each acid–base reaction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





