
Concept explainers
Interpretation:
The Kekulé’s structure of benzene needs to be drawn and the reason why this doesn’t represent actual structure needs to be explained.
Concept introduction:
Benzene is an
Explanation of Solution
A sensible structure of benzene was proposed by Kekulé. According to him, the carbons are arranged in hexagon with alternate single and double bonds present between them. Each carbon is attached to one hydrogen atom via single bond. The proposed structure of Kekulé is:
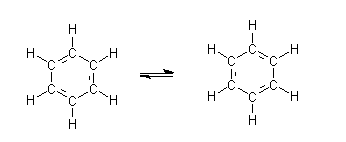
It does not represent correct structure of benzene as:
- It suggests two different type of carbon-carbon bonds in benzene that is, single and double carbon-carbon bond but in benzene all the bonds are exactly same and benzene is planar molecule.
- Due to presence of three double bond, it can be expected that benzene undergoes addition reaction as those of
alkenes. But benzene rarely undergoes addition reaction whereas it undergoes substitution reactions. - The real structure of benzene is very stable in comparison to the structure of benzene proposed by Kekulé.
Chapter 21 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Chemistry: The Central Science (14th Edition)
Chemistry: A Molecular Approach
General Chemistry: Principles and Modern Applications (11th Edition)
Organic Chemistry (9th Edition)
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





