
Interpretation:
Using given table, a statement needs to be constructed that explains the relationship between number of carbon atoms of the
Concept introduction:
The temperature at which a liquid substance becomes gas is said to be boiling point. At this temperature, the pressure surrounding the liquid and vapor pressure of the liquid becomes equal.
Explanation of Solution
Given:
The table:
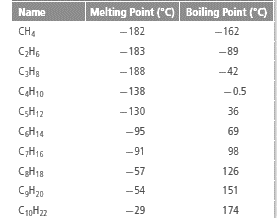
From the given table it can be observed that the methane containing one carbon atom has lowest boiling point, 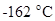 and decane containing 10 carbon atoms has highest boiling point,
and decane containing 10 carbon atoms has highest boiling point, 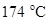 The increase in boiling point is observed with increase in number of carbon atoms in alkanes. The intermolecular force of attraction between the alkanes is Van der Waals attraction which increases with increase in surface area of the interacting electron clouds and hence, the large molecule is decane has greater surface area of electron clouds and have greater boiling point.
The increase in boiling point is observed with increase in number of carbon atoms in alkanes. The intermolecular force of attraction between the alkanes is Van der Waals attraction which increases with increase in surface area of the interacting electron clouds and hence, the large molecule is decane has greater surface area of electron clouds and have greater boiling point.
Chapter 21 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: The Central Science (13th Edition)
Organic Chemistry
Chemistry: The Central Science (14th Edition)
Chemistry: Structure and Properties (2nd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





