
Interpretation:
The geometric arrangement of the bonds around the carbon atoms in
Concept introduction:
The geometric shape of the molecule is predicted by VSEPR theory (Valence shell electron pair repulsion theory) from electron pairs surrounding the central atom of the molecule.
Answer to Problem 24SSC
According to VESPR the shape of the molecule:
Alkane is tetrahedral.
Alkene is trigonal planar.
Alkyne is linear.
Explanation of Solution
The geometric arrangement of the bonds around the carbon atoms in alkanes, alkenes, and alkynes are determined as:
- The structure of alkanes has single bond between carbon and hydrogen atoms. So, in alkane there is 4 bonded atoms and no lone pair across the carbon atom and thus, according to VESPR the shape of the molecule will be tetrahedral.
- The structure of alkenes has double bond between two carbon atoms. So, in alkene there is 3 bonded atoms and no lone pair across the carbon atom and thus, according to VESPR the shape of the molecule will be trigonal planar shape.
- The structure of alkynes has triple bond between two carbon atoms. So, in alkyne there is 2 bonded atoms and no lone pair across the carbon atom and thus, according to VESPR the shape of the molecule will be linear.
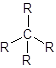
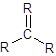
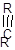
Chapter 21 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Organic Chemistry (9th Edition)
Organic Chemistry (8th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry: The Central Science (13th Edition)
Inorganic Chemistry
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





