
Interpretation:
The highlights about a molecule by four molecular models needs to be identified.
Concept introduction:
A scale model which represents the arrangement of atoms in the molecule is said to be molecular model.
Explanation of Solution
The four molecular models are:
- Molecular formula: It represents
chemical symbols of the elements in the molecule and the number of atoms of each element is represented by subscript.
For example:
- Structural formula: It consists of symbols of the elements and lines for the
chemical bonds .
For example: The structural formula of methane is:
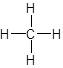
- Ball-and-stick model: It displays the three-dimensional position of the atoms and the chemical bonds present in a molecule. Differently colored spheres are used to represent atoms and are connected by rods that represents chemical bonds.
For example: The ball-and-stick model of methane is:
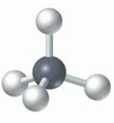
The black sphere represents carbon atom and grey balls represents hydrogen atoms.
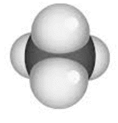
- Space-filling model: The relative space that each atom exerts it self in is represented by space-filling model.
For example: The space-filling model of methane is:
Chapter 21 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry (3rd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: Structure and Properties
Organic Chemistry (9th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





