
Interpretation:
The other functional group is identified in Capsaicin other than phenol.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Amide:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with nitrogen is called as amide.
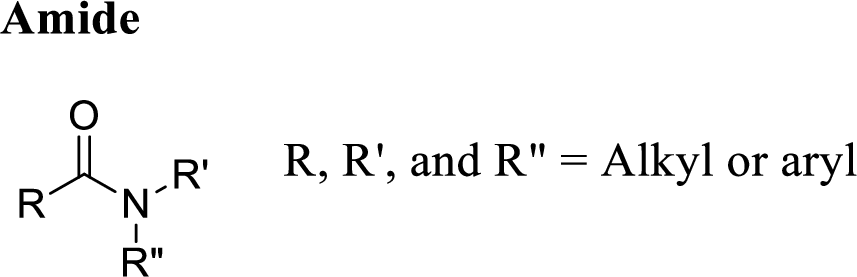
Alkenes are a class of hydrocarbons. The carbon-carbon double bond is called as alkenes and it is also called as olefins.
Ether:
The aliphatic or
Example is given below
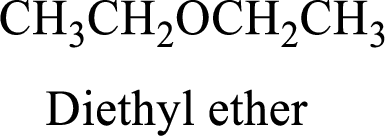
Trending nowThis is a popular solution!

Chapter 21 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Amide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forward1. Which of the following statements is true? I. Aldehydes and ketones both contain a hydroxyl group. II. The names for aldehydes and ketones are derived from the name of the longest carbon chain that contains the carbonyl group. III. The aldehyde and ketone with a molecular formula of C3H6O are constitutional isomers. IV. 2-Propanone is immiscible in water. A. I & II B. II & II C. I & III D. I & IV 2. Whicb of the following is the correct IUPAC name of the structure below?arrow_forward2. Oxybenzone and 2-ethylhexyl-p-methoxycinnamate are UV absorbing compounds used in sunscreens. Identify and encircle the functional groups in these compounds. a. Oxybenzone Ö HÓ b. 2-ethylhexyl-p-methoxycinnamatearrow_forward
- What are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. acetic acid and toluene b. phenylic acid and ethanol C. ethanoic acid and benzene d. benzoic acid and ethanol e. phenylic acid and methanol O a O barrow_forward1. An amine is characterized by what functional group? a. -CO2CH3 b. -NH2 d. -CHO e. -OH c. -CO2H 2. Which of the follow ing is an akohol? a. NaOH d. CH CH 20OH b. H3CNH2 c. H3C-C-CH3 с. 3. When two alcohols undergo a self condensation, what is formed? a. liquid alcohol b. a ketone d. an aklehyde an ether e. с. an esterarrow_forward1. what priority functional group of ff organic compound? a. carboxyl b. hydroxide c. hydroxyl d. carbonyl 2. what group does the ff organic compound belong? a. azo b. diazo c. aromatic d. organosulfur 3. what group does the ff organic compound belong? a. azo b. amines c. diazo d. nitrilesarrow_forward
- True or falsearrow_forwardPolyphenols are compounds found in many fruits, vegetables, tea, and wine. Predict the solubility of this polyphenol, gallic acid, in water.arrow_forward1. Which is water-soluble? Why? 2. What is another property which will make each one different from the others?arrow_forward
- For embalming purposes, phenol has the properties of preserving, disinfecting, and ___ . cauterizing cut tissues adding a pink glow to the body adding a nice fragrance anticoagulantarrow_forward16. An atom or group of atoms that can give organic compounds distinct chemical and physical properties. 21. When a compound with the general formula R-COOH loses a proton, the product that remains is described with this term. Its general formula is R-COO- 24. A class of organic compounds in which three or more carbons form a ring structure. All of the carbon-to-carbon bonds are single bonds in this family of compounds.arrow_forward4. The organic starting materials for the preparation of an ester could be C. a ketone and alcohol A an acid and alcohol B. water and oxygen D. alkane and aldehydearrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



