
Concept explainers
Interpretation: (a) The Lewis structures of the alkane having seven carbon chain and one methyl as a side chain have to be drawn. (b) The chiral carbon containing structures among them are to be identified.
Concept introduction:
Structural Isomers are the structure of a molecule with same molecular formula but have different arrangements of bonds and atoms. For a single molecular formula of
A chiral carbon is the carbon atom which is substituted to four different groups. Due to presence of chiral carbon a molecule become chiral and it gives optical isomers also that is
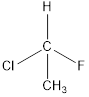
All the substituents are different hence is is a chiral carbon and so molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Pac Mindlink Owlv2 (24 Months) Chemistry & Chemical Reactivity 9e
- In the structure of 4-isopropyl-2,4,5-trimethylheptane, identify the primary, secondary, tertiary, and quaternary carbons. Use the abbreviations: p is primary, s is secondary, t is tertiary and q is quarternary.arrow_forwardDraw three cycloalkanes which are structural (constitutional) isomers of C6H12. At least one of these structures must be able to show cis/trans isomerism. Mark this example with an asterisk.arrow_forwardexplain structural isomerism in aliphatic alkanes and geometric isomerism in alkenes. Describe structural isomerism and the different types that exist. With the aid of diagrams use the molecular formulae C5H12 and C5H10 to explain structural isomerism in aliphatic alkanes and alkenes. Describe geometric isomerism. With the aid of diagrams use the molecular formula C5H10 to explain geometric isomerism in alkenes.arrow_forward
- There are 11 structures (ignoring stereoisomerism) with the formula C4H8O that have no carbon branches. Draw the structures and identify the functional groups in each.arrow_forwardThe empirical formula of X is C3H5CIO If X is an optically alkanal, give the structural formula of X and draw this pair of enantiomers.Structural formula of X : If X has a C=C bond and a saturated OH group (i.e. The OH group does not attach to the C=C), give the structural formula of X if X shows cis-trans isomerism. Draw the pair of cis-trans isomers.Structural formula of X :arrow_forward1-bromo-3-ethylcyclopentane exhibits geometrical isomerism. Draw structures of the two geometrical isomersarrow_forward
- how many alkanes of the formula c6h14 have quaternary carbon atom?arrow_forwardMineral oil and Vaseline are both mixtures of alkanes, but mineral oil is a liquid at room temperature and Vaseline is a solid. Which product is composed of alkanes that contain a larger number of carbon atoms? Explain your choice.arrow_forwardExplain how the boiling point of substances varies in a homologous series of alkanes? Provide examples.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





