
Pac Mindlink Owlv2 (24 Months) Chemistry & Chemical Reactivity 9e
9th Edition
ISBN: 9781305389762
Author: Kotz
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 23, Problem 83GQ
Interpretation Introduction
Interpretation:
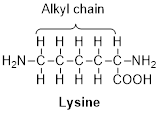
The group that is replaced to convert lysine into cadaverine is to be stated.
Concept introduction:
Lysine is one of the essential amino acid from
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the full structure of an amine which is an isomer of ethylamine? What is the isomer called? Is it primary, secondary or tertiary?
Aspartame, an artificial sweetener used in Equal® and diet beverages, has the following structure. Identify the functional groups in
Aspartame.
II
NH, O
OH
O 1= ester; I| = amide; III = amine
O 1 = ester; I| = amine; III = amide
O1= ether; I| = amide; III = amine
O 1 = ether; I| = amine; III = amide
O 1 = anhydride; Il = ketone; I|| = amine
19.
Which statement about structural (constitutional isomers) is CORRECT?
A) Structural (constitutional) isomers are two compounds with the same molecular
formula and the same structure.
B) Structural (constitutional) isomers are compounds which always have the same
carbon chain length.
C) Structural (constitutional) isomers are compounds in which the functional group will
always be in the same position.
D) Structural (constitutional) isomers are compounds that contain the same number of
atoms of each element but the specific arrangement of the atoms differs.
20.
The following two compounds are
CH3
CH3
Br
Br
A) Constitutional isomers
B) Conformational isomers
C) Geometric isomers
D) Enantiomers
Chapter 23 Solutions
Pac Mindlink Owlv2 (24 Months) Chemistry & Chemical Reactivity 9e
Ch. 23.2 - (a) Draw the nine isomers having the formula...Ch. 23.2 - Prob. 2CYUCh. 23.2 - There are 17 possible alkene isomers with the...Ch. 23.2 - Prob. 4CYUCh. 23.2 - Aniline, C6H5NH2, is the common name for...Ch. 23.2 - What is the systematic name for this alkane? (a)...Ch. 23.2 - Prob. 2RCCh. 23.2 - Prob. 3RCCh. 23.2 - Prob. 4RCCh. 23.2 - 5. How many isomers are possible for C6H4(CH3)Cl,...
Ch. 23.3 - Draw the structure of 1-butanol and alcohols that...Ch. 23.3 - Prob. 1RCCh. 23.3 - Prob. 2RCCh. 23.3 - What is the hybridization of nitrogen in...Ch. 23.3 - Prob. 4RCCh. 23.4 - (a) Name each of the following compounds and its...Ch. 23.4 - Prob. 1RCCh. 23.4 - Prob. 2RCCh. 23.4 - Prob. 3RCCh. 23.4 - Prob. 4RCCh. 23.4 - Prob. 1QCh. 23.4 - Prob. 2QCh. 23.4 - Prob. 3QCh. 23.5 - Kevlar is a well-known polymer that is now used to...Ch. 23.5 - Prob. 1QCh. 23.5 - Prob. 2QCh. 23.5 - Prob. 3QCh. 23.5 - Prob. 1RCCh. 23.5 - What is the atom economy for the reaction of...Ch. 23.5 - Prob. 5QCh. 23.5 - If drinking from a polycarbonate bottle, does a 15...Ch. 23.5 - Assume you weigh 156 lb. How much BPA do you...Ch. 23.5 - Prob. 8QCh. 23 - Prob. 1PSCh. 23 - What is the molecular formula for an alkane with...Ch. 23 - Prob. 3PSCh. 23 - Prob. 4PSCh. 23 - One of the structural isomers with the formula...Ch. 23 - Prob. 6PSCh. 23 - Prob. 7PSCh. 23 - Give the systematic name for the following alkane....Ch. 23 - Draw the structure of each of the following...Ch. 23 - Draw structures for the following compounds. (a)...Ch. 23 - Prob. 11PSCh. 23 - Prob. 12PSCh. 23 - Draw the structure of the chair form of...Ch. 23 - Prob. 14PSCh. 23 - Prob. 15PSCh. 23 - Prob. 16PSCh. 23 - Prob. 17PSCh. 23 - Prob. 18PSCh. 23 - Prob. 19PSCh. 23 - What structural requirement is necessary for an...Ch. 23 - A hydrocarbon with the formula C5H10, can be...Ch. 23 - Prob. 22PSCh. 23 - Prob. 23PSCh. 23 - Prob. 24PSCh. 23 - The compound 2-bromobutane is a product of...Ch. 23 - The compound 2,3-dibromo-2-methylhexane is formed...Ch. 23 - Prob. 27PSCh. 23 - Prob. 28PSCh. 23 - Prob. 29PSCh. 23 - Prob. 30PSCh. 23 - Prob. 31PSCh. 23 - Give the systematic name for each of the following...Ch. 23 - Prob. 33PSCh. 23 - Write an equation for the preparation of...Ch. 23 - Prob. 35PSCh. 23 - Prob. 36PSCh. 23 - Prob. 37PSCh. 23 - Prob. 38PSCh. 23 - Prob. 39PSCh. 23 - Name the following amines: (a) CH3CH2CH2NH2 (b)...Ch. 23 - Draw structural formulas for the four possible...Ch. 23 - Prob. 42PSCh. 23 - Prob. 43PSCh. 23 - Prob. 44PSCh. 23 - Prob. 45PSCh. 23 - Prob. 46PSCh. 23 - Prob. 47PSCh. 23 - Prob. 48PSCh. 23 - Prob. 49PSCh. 23 - Prob. 50PSCh. 23 - Give the structural formula and systematic name...Ch. 23 - Prob. 52PSCh. 23 - Prob. 53PSCh. 23 - Prob. 54PSCh. 23 - Prob. 55PSCh. 23 - Prob. 56PSCh. 23 - Prob. 57PSCh. 23 - Prob. 58PSCh. 23 - Prob. 59PSCh. 23 - Prob. 60PSCh. 23 - Identify the functional groups in the following...Ch. 23 - Prob. 62PSCh. 23 - Prob. 63PSCh. 23 - Prob. 64PSCh. 23 - Prob. 65PSCh. 23 - Prob. 66PSCh. 23 - Prob. 67GQCh. 23 - Prob. 68GQCh. 23 - Prob. 69GQCh. 23 - Prob. 70GQCh. 23 - Prob. 71GQCh. 23 - Prob. 72GQCh. 23 - Prob. 73GQCh. 23 - Write equations for the following reactions,...Ch. 23 - Prob. 75GQCh. 23 - Prob. 76GQCh. 23 - Draw the structure of each of the following...Ch. 23 - Prob. 78GQCh. 23 - Prob. 79GQCh. 23 - Draw structural formulas for possible isomers with...Ch. 23 - Prob. 81GQCh. 23 - Prob. 82GQCh. 23 - Prob. 83GQCh. 23 - Prob. 84GQCh. 23 - Prob. 85GQCh. 23 - Prob. 86GQCh. 23 - Draw the structure of glyceryl trilaurate, a fat....Ch. 23 - Prob. 88GQCh. 23 - Prob. 89GQCh. 23 - Prob. 90GQCh. 23 - Prob. 91GQCh. 23 - There are three ethers with the formula C4H10O....Ch. 23 - Review the opening photograph about chocolate...Ch. 23 - Prob. 94GQCh. 23 - Prob. 95ILCh. 23 - Prob. 96ILCh. 23 - Prob. 97ILCh. 23 - Prob. 98ILCh. 23 - Prob. 99ILCh. 23 - Prob. 100ILCh. 23 - Prob. 101ILCh. 23 - Prob. 102ILCh. 23 - Prob. 103ILCh. 23 - Prob. 104ILCh. 23 - Prob. 105ILCh. 23 - Prob. 106ILCh. 23 - Prob. 107SCQCh. 23 - Prob. 108SCQCh. 23 - Prob. 109SCQCh. 23 - Prob. 110SCQCh. 23 - Prob. 111SCQCh. 23 - Prob. 112SCQCh. 23 - Prob. 113SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Beeswax contains this compound: Identify what type of compound this is. Write the structural formulas of the compounds produced by the hydrolysis of this compound.arrow_forwardGlutathione is a tripeptide whose function is to destroy harmful oxidizing agents in the body. Oxidizing agents are thought to be responsible for some of the effects of aging and to play a causative role in cancer. Glutathione removes oxidizing agents by reducing them. In the process, glutathione is oxidized, resulting in the formation of a disulfide bond between two glutathione molecules. An enzyme subsequently reduces the disulfide bond, returning glutathione to its original condition so it can react with another oxidizing agent.a. What amino acids make up glutathione? b. What is unusual about glutathione’s structure?arrow_forwardTwo compounds given below had the following IUPAC name and structural formula 2-aminopropanoic acid (alanine) CH3CHNH2COOH 1,2-dibromocyclopentane C3H6CHBrCHBr (i) What type of isomerism is possible in the 2 organic compounds? (ii) Draw all the pairs of possible isomers and name them.arrow_forward
- 16. Given the organic functional group: II -C-H Which class of organic compounds has molecules with this functional group? (1) aldehydes (2) esters (3) ketones (4) organic acidsarrow_forwardAs we’ve discussed, a peptide bond is made when amino group of one amino acid combines with the carboxylic acid group of another amino acid (releasing a water molecule in the process). The C-N bond formed in this process is called a peptide bond. Peptide bonds have a few properties that might be unexpected. a) One property is that the molecular geometries around the C and N atom in the peptide bond are generally planar with bond angles of approximately 120 degrees. Provide an explanation for why the peptide bond would have this property, using Lewis structures, VSEPR theory and/or valence bond theory as appropriate.arrow_forwardTRUE OR FALSE (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one —OH group, a secondary alcohol has two —OH groups, and a tertiary alcohol has three —OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic…arrow_forward
- How do amines differ from analogous alcohols in (a) odor,(b) basicity, and (c) boiling point?arrow_forwardExplain the meaning of the word “organic” when used to describe certain foods. How is it related to the term “organic” when used with organic chemistry?arrow_forwardHydrogen peroxide is not only an antiseptic but also an oxidizing agent. The following equation shows the reaction of hydrogen peroxide with acetaldehyde to give acetic acid: C2H4O(l) + H2O2(l) → C2H4O2(l) + H2O(l) In this reaction, which species is oxidized and which species is reduced? Which species is the oxidizing agent and which species is the reducing agent?arrow_forward
- Organic Compounds: "Amino acids vary only by R group" What do they mean by R group/what is the R group?arrow_forwardThe following is the structure of naproxen, a common nonsteroidal anti inflammatory drug What type of structural formula is this? O ||.⁰⁰ OHarrow_forwardProf Adam and his colleagues synthesized compound E, with the molecular formula CSH1CI. They wanted to identify the chemical properties of this compound by conducting a series of chemical tests. Compound E produced compound F when it reacted with sodium hydroxide in the presence of water and reflux. In the presence of ultraviolet light, compound G reacts with halogen gas to form compound E. Compound E is formed when compound H undergoes addition reactions involving hydrogen chloride. When compound E reacts with aqueous ammonia in the presence of heat, compound I is formed. Draw the possible skeletal structures of compound F to I. Identify the IUPAC nomenclature of compounds F, G, H, and I.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY