
Concept explainers
Interpretation:
The structures of
Concept introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry).IUPAC name consists of three parts, namely Prefix, suffix and root word.
Prefix- Represents the substituent present in the molecule and its position in the root name.
Suffix- Denotes the presence of functional group if any in the molecule. It can be an
Root word - Represents the longest continuous carbon skeleton of the organic molecule.
A chiral carbon is the carbon atom which is substituted to four different groups. Due to presence of chiral carbon a molecule become chiral and it gives optical isomers also that is
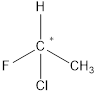
All the substituents are different hence it is a chiral carbon and so molecule.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Pac Mindlink Owlv2 (24 Months) Chemistry & Chemical Reactivity 9e
- Organic Chemistry HW: CANNOT BE HAND DRAWN 2,6-dimethyloct-2-ene Provide a detailed typed explanation of Stereoisomers show the expanded structure of your molecule. Calculate the maximum number of possible stereoisomers of your molecule using the following formula: Maximum number of possible stereoisomers = 2n (where n= the number of chiral carbons in your molecule). This calculation does not include E- or Z- isomers for any compounds containing double bonds Type or using a computer program "draw" the possible stereoisomers of the molecule. Note that E-, Z- isomers of each stereoisomer are also possible and would not be accounted for by the formula above; draw any E- or Z- isomers.arrow_forwardThis molecule (is or is not) a reducing sugar. The sugar molecule is attached to the alcohol at carbon (just use a number) in the steroid structure. The lactone group is attached to carbon (just use a number) in the steroid structure. There are (just use a number) chiral carbons in this molecule. CH3 HỌ но. но, H A ÕH но ÕH CH3 • 8H2O ОН ОНarrow_forwardHow many chiral carbons are present in the structural formula for the open-chain form of d-mannose? Draw the structure of the six-member-ring form of this molecule.arrow_forward
- Part A: Celecoxib Part B: O-cresolarrow_forwardHow many chiral carbon atoms are in this structure? Br OH ОН CIarrow_forwardFructose and glucose both contain six carbon atoms. Why do the cyclic forms of fructose have a five-membered ring instead of the six-membered ring found in the cyclic forms of glucose? Draw the skeletal structures.arrow_forward
- Consider the molecule N-ethyl-N-methyl-3-iodo-4-methyl hexanamide. Draw it and give the two reactants that would produce this product. Name them and draw their skeletal structures.arrow_forwardThe prevalence of these organic substituents would indicate that a bio-molecule is a lipid? A) amide B) disulfide C) ester D) alcohol E) lots of hydrocarbons in rings or chains The prevalence of this organic functional group would indicate that a bio-molecule is a carbohydrate. A) amide B) disulfide C) ester D) alcohol E) lots of hydrocarbons in rings or chainsarrow_forwardIndicate whether or not each of the following statements is TRUE or FALSE. Justify your answer with ONE or TWO sentences. i. Lipids are considered to be more soluble in water than in other solvents. ii. The many -OH groups present in carbohydrates such as glucose make them quite insoluble in water and blood. ii. Carbohydrate structure shows that water molecules are connected to carbon atoms. iv. Hydrocarbons contain carbon, hydrogen and oxygen. A monosaccharide can be hydrolyzed to smaller units. V.arrow_forward
- Indicate whether or not each of the following statements is TRUE or FALSE. Justify your answer with ONE or TWO sentences. i. Lipids are considered to be more soluble in water than in other solvents. ii. The many -OH groups present in carbohydrates such as glucose make them quite insoluble in water and blood. iii. Carbohydrate structure shows that water molecules are connected to carbon atoms. iv. Hydrocarbons contain carbon, hydrogen and oxygen. v. A monosaccharide can be hydrolyzed to smaller units.arrow_forwardExplain the Biodegradable Polymers ?arrow_forwardClassify the functional groups in the following steroid (choose all that apply).CHOOSE ALL THAT APPLY a. alkyne b. secondary alcohol c. aldehyde d. aromatic e. alkene f. ester g. ketone h. phenol i. tertiary alcohol j. primary alcohol k. carboxylic acidarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College DivChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College DivChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





