
Concept explainers
(a)
Interpretation:
Nitrogen atom that is a more basic among the two nitrogen atoms present in epibatidine has to be explained.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
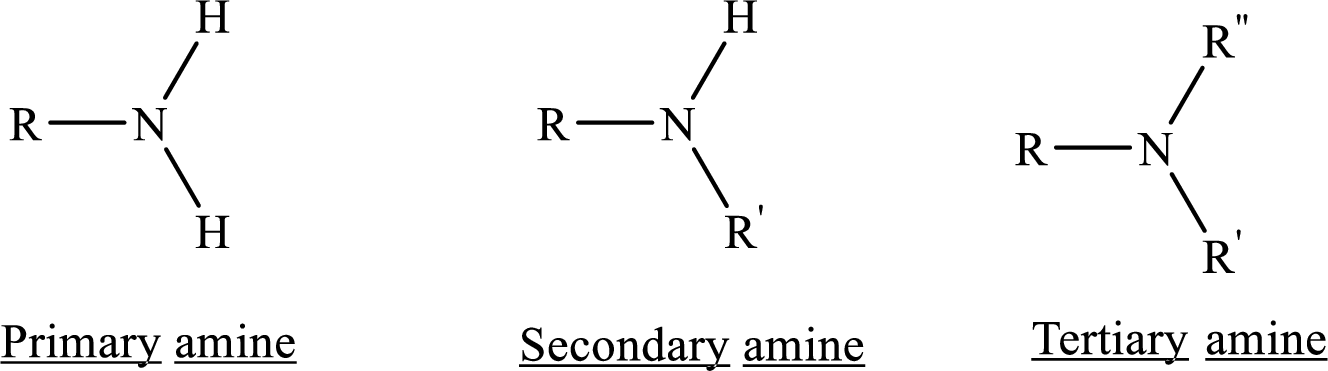
According to the explanations by Lewis, if a species donates an electron pair then it is a Lewis base whereas if a species receives an electron pair, then it is Lewis acid.
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
(b)
Interpretation:
Chiral centers in epibatidine have to be marked.
Concept Introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
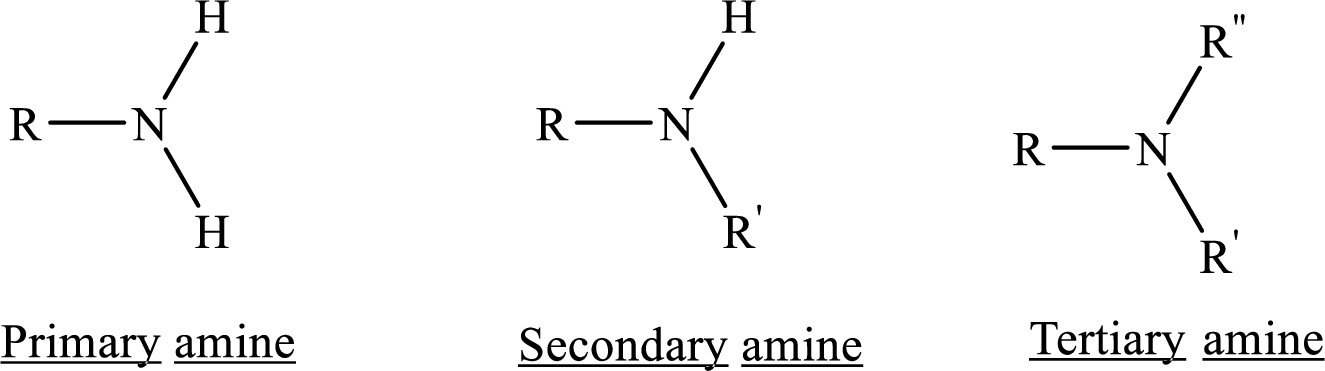
Chirality: It refers to an atom in a molecule that contains four different substituents.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Phenacetin, a substance formerly used in over-the-counter headache remedies, has the formula C10H13NO2. Phenacetin is neutral and does not dissolve in either acid or base. When warmed with aqueous NaOH, phenacetin yields an amine, C8H11NO, whose 1H NMR spectrum is shown. When heated with HI, the amine is cleaved to an aminophenol, C6H7NO. What is the structure of phenacetin, and what are the structures of the amine and the aminophenol?arrow_forwardCiprofloxacin is a member of the fluoroquinolone class of antibiotics.(a) Which of its rings are aromatic?(b) Which nitrogen atoms are basic?(c) Which protons would you expect to appear between d 6 and d 8 in the proton NMR spectrum?arrow_forwardWhat is the conjugate acid of acetamide and its pka value?arrow_forward
- The shrub ma huang (Section 5.4A) contains two biologically activestereoisomers—ephedrine and pseudoephedrine—with two stereogeniccenters as shown in the given structure. Ephedrine is one component ofa once-popular combination drug used by body builders to increaseenergy and alertness, whereas pseudoephedrine is a nasaldecongestant.a.) Draw the structure of naturally occurring (−)-ephedrine, which has the1R,2S configuration.b.) Draw the structure of naturally occurring (+)-pseudoephedrine, whichhas the 1S,2S configuration.c.) How are ephedrine and pseudoephedrine related?d.) Draw all other stereoisomers of (−)-ephedrine and (+) pseudoephedrine, and give the R,S designation for all stereogeniccenters.e.) How is each compound drawn in part (d) related to (−)-ephedrine?arrow_forwardFollowing is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker with high selectivity for airway smooth muscle receptors. Q.Suggest a structural relationship between carbuterol and ephedrinearrow_forwardWhen a compound with molecular formula C11H14O2 undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following 1H NMR spectrum. Identify the compound.arrow_forward
- Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia. Is that true or false?arrow_forwardHypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that produces seizures, coma, and sometimesdeath in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?arrow_forwardWhen a compound with molecular formula C11H14O2 undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following1H NMR spectrum. Identify the compound.arrow_forward
- Compound 1 is an anticoagulant that is extracted from, among other plants, the sweetclover plant, Melilotus Officinalis. However, the compound is moderately toxic and alsofinds use as a rodenticide. Name this compoundarrow_forwardPlace the binders below in descending order of acidity (pi) and justify your choice: CH3CN; (C2H5)2O; PCl3; As(C6H5)3; (C2H5)3Narrow_forwardRank each set of compounds in order of increasing basicity.pyrrole, imidazole, 3-nitropyrrolearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

