
(a)
Interpretation:
The synthesis of fexofenadin has to be shown.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
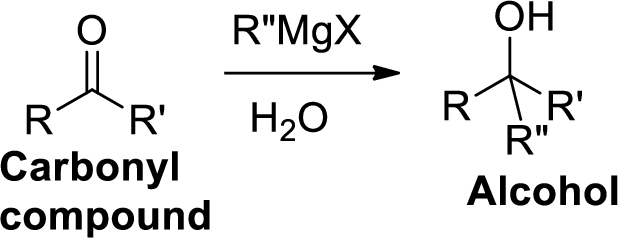
(b)
Interpretation:
The possible stereoisomer’s has to be shown if the product is chiral.
Concept introduction:
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Organic Chemistry
- What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.arrow_forwardThe two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides.Show how these techniques can be used to accomplish the following syntheses.(a) benzoic acid S benzylamine (b) benzaldehyde S benzylamine(c) pyrrolidine S N@ethylpyrrolidine (d) cyclohexanone S N@cyclohexylpyrrolidine(e) HOOC¬(CH2)3 ¬COOH S pentane@1,5@diamine (cadaverine)arrow_forwardThe catalog prices for all reagents used to make Phenacetin are shown: p-ethoyxanaline: 100 grams, $26.00 (Weight used: 2.5 grams) Acetic Anhydride: 1000 grams, $39.50 (Weight used: 2.2 grams, Density: 1.08) Hydrochloric Acid: 2500 mL, $30.20 (Weight used: 1.53 grams, Density: 1.02) Sodium Acetate Trihydrate: 500 grams, $30.50 (Weight used: 3.0 grams) Calculate the cost of each reagent.arrow_forward
- Predict the chemical name of compound B a. p-chlorobenzylamine b. 4-bromoaniline c. benzylamine d. p-chloronitrile e. p-chlorobenzaldehyde 2. What is the reaction name for the chemical transformation of A to B a. reductive amination b. catalytic reduction c. carbonyl dehydration d. Hofmann elimination e. Aldehyde rearrangementarrow_forwardFollowing are structural formulas for two more widely used sulfonylurea hypoglycemic agents.Show how each might be synthesized by converting an appropriate amine to a carbamic ester and then treating the carbamate with the sodium salt of a substituted benzenesulfonamide.arrow_forwardShow how to synthesize the following amines from the indicated starting materials below. Each of the final products may require several sequential steps. Draw the structure for each of the steps and show the reagents required. Starting material Final product e) 5-methylhexan-2-one 1-amino-2,5-dimethylhexan-2-ol f) 1-iodopropane tetrapropylammonium iodide g) 4-ethyl-3-methylhexan-1-ol 3-ethyl-2-methylpentan-1-aminearrow_forward
- 1. What are the chemical hazards of both aniline and acetic anhydride? 2. What are the medicinal and industrial uses/application of acetanilide? 3. What was the undesirable effect of acetanilide that made it toxic? 4. How does acetanilide produce an analgesic effect in the body?arrow_forwardAn amine of unknown structure contains one nitrogen and nine carbon atoms. The 13C-NMR spectrum shows only five signals, all between 20 and 60 ppm. Three cycles of Hofmann elimination sequence [(1) CH3I; (2) Ag2O, H3O; (3) heat] give trimethylamine and 1,4,8-nonatriene. Propose a structural formula for the amine.arrow_forwardWhy is methyl salicylate so easily absorbed through the skin?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


