
(a)
Interpretation:
Synthesis of 3-nitrophenol has to be proposed using 3-nitroaniline as starting material.
Concept Introduction:
Conversion of
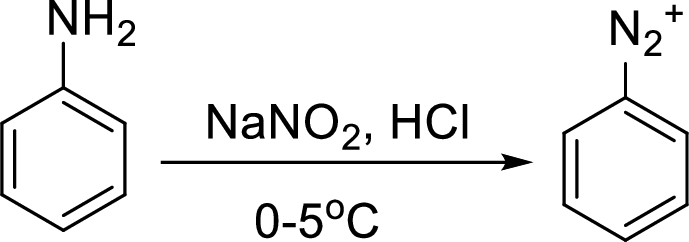
Aromatic amines converted to arenediazonium salt by reacting with
(b)
Interpretation:
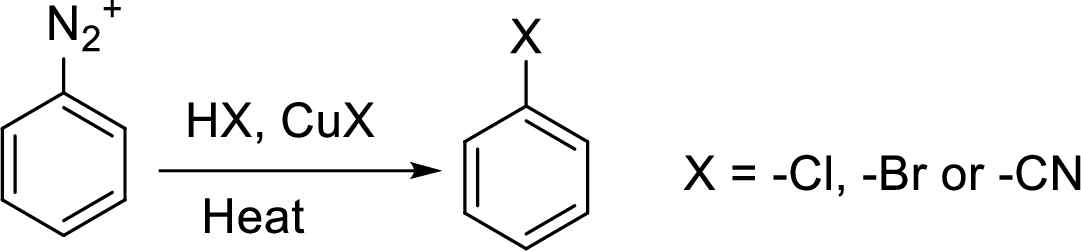
Synthesis of 3-bromo nitrobenzene has to be proposed using 3-nitroaniline as starting material.
Concept Introduction:
Reaction of a primary aromatic amine with sodium nitrite:
The reaction of a primary aromatic amine with sodium nitrite in presence of aqueous
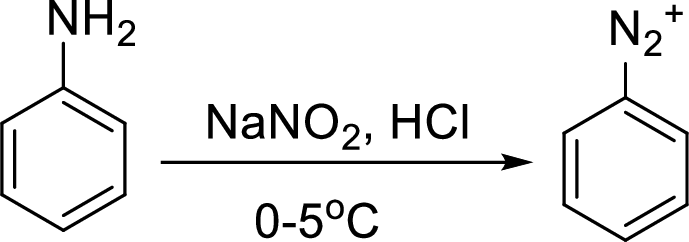
Sandmeyer reaction: It reaction type of organic reaction where the diazonium group in an arenediazonium salt gets replaced by
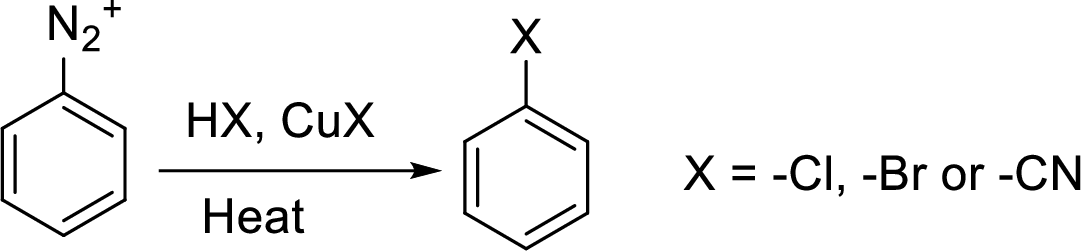
Hydrogenation:
Hydrogenation means the addition of hydrogen molecules to the unsaturated compound which makes them saturated hydrocarbon in the presence of catalyst.
(c)
Interpretation:
Synthesis of 1,3-dihydroxybenzene has to be proposed using 3-nitroaniline as starting material.
Concept Introduction:
Reaction of a primary aromatic amine with sodium nitrite:
The reaction of a primary aromatic amine with sodium nitrite in presence of aqueous
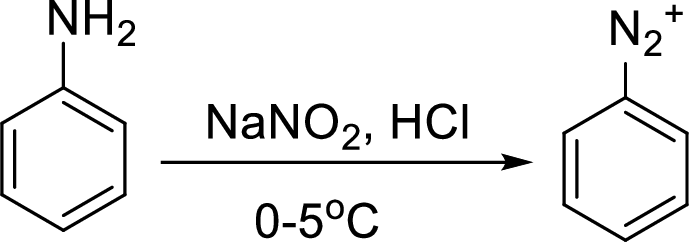
Hydrogenation:
Hydrogenation means the addition of hydrogen molecules to the unsaturated compound which makes them saturated hydrocarbon in the presence of catalyst.
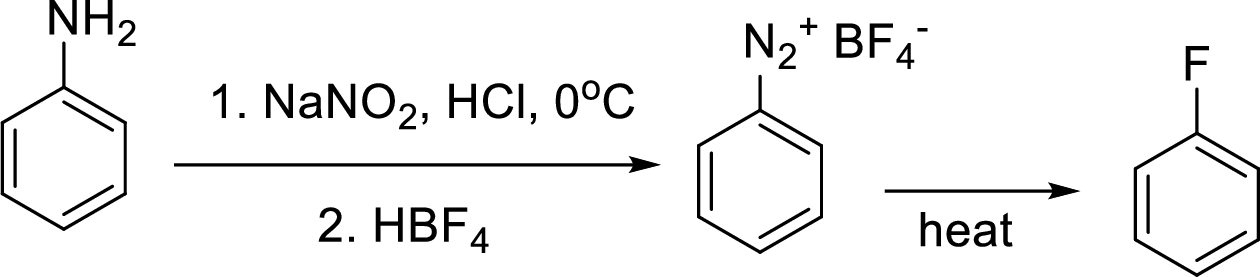
(d)
Interpretation:
Synthesis of 3-fluoroaniline has to be proposed using 3-nitroaniline as starting material.
Concept Introduction:
Schiemann reaction: It is a method used to introduce fluorine into an aromatic ring. The reaction of a primary aromatic amine with sodium nitrite in presence of aqueous
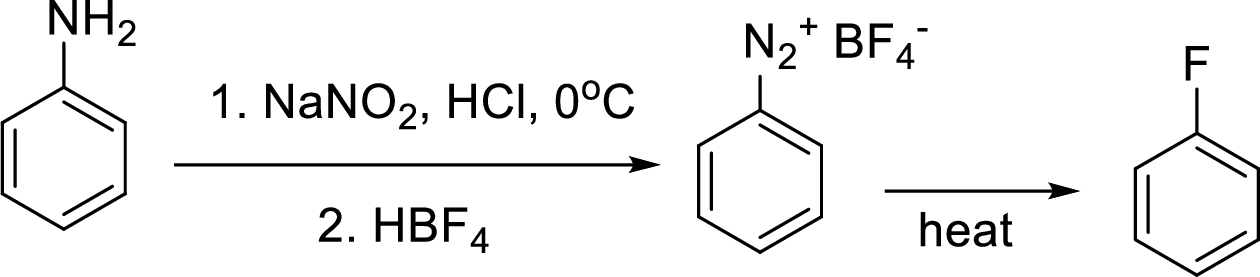
(e)
Interpretation:
Synthesis of 3-fluorophenol has to be proposed using 3-nitroaniline as starting material.
Concept Introduction:
Conversion of aromatic amines to phenol:
Aromatic amines converted to arenediazonium salt by reacting with
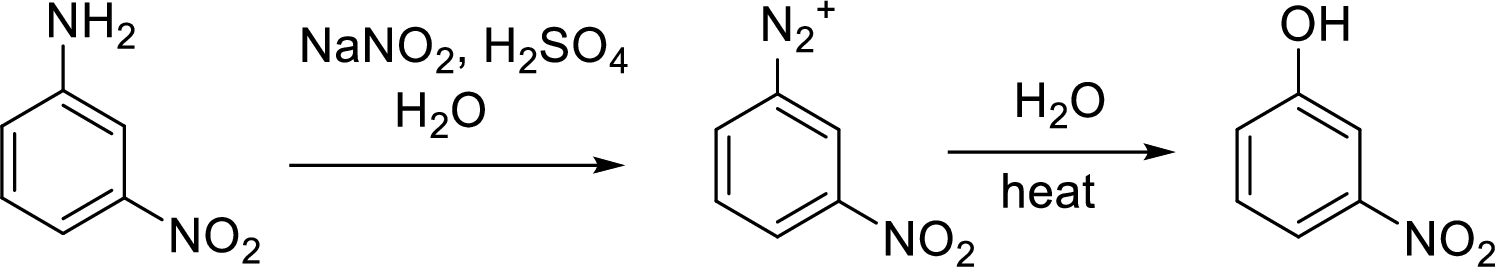
Schiemann reaction: It is a method used to introduce fluorine into an aromatic ring. The reaction of a primary aromatic amine with sodium nitrite in presence of aqueous
(f)
Interpretation:
Synthesis of 3-hydroxybenzonitrile has to be proposed using 3-nitroaniline as starting material.
Concept Introduction:
Hydrogenation:
Hydrogenation means the addition of hydrogen molecules to the unsaturated compound which makes them saturated hydrocarbon in the presence of catalyst.
Reaction of a primary aromatic amine with sodium nitrite:
The reaction of a primary aromatic amine with sodium nitrite in presence of aqueous
Sandmeyer reaction: It reaction type of organic reaction where the diazonium group in an arenediazonium salt gets replaced by
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Reaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0 °C, followed by treatment with N,N-diethylaniline.arrow_forwardShow how you would use extractions with a separatory funnel to separate a mixture of the following compounds: benzoicacid, phenol, benzyl alcohol, anilinearrow_forwardThe following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forward
- Benzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.arrow_forwardA compound of formula C11H16N2 gives the IR, 1H NMR, and 13C NMR spectra shown. The proton NMR peak at δ 2 disappears on shaking with D2 Propose a structure for this compound, and show how your structure accounts for the observed absorptions.arrow_forwardThe compound acetophenone has a very similar molar mass to that of benzoic acid and benzamide. However, acetophenone has a much lower m.p. (20 °C) than both such that, by contrast, it is a liquid at room temperature. By considering intermolecular forces and comparing functional group structure, account for this big difference in physical properties.arrow_forward
- Place the binders below in descending order of acidity (pi) and justify your choice: CH3CN; (C2H5)2O; PCl3; As(C6H5)3; (C2H5)3Narrow_forwardPropose a structural formula for compound A, C5H13N, given its IR and calculated 1H-NMR spectra.arrow_forwardAssume that you have samples of the following two compounds, both with formula C7H8O. Both compounds dissolve in ether, but only one of the two dissolves in aqueous NaOH. How could you use this information to distinguish between them?arrow_forward
- The sex attractant of the housefly has the formula C23H46. When treated with warm potassium permanganate, this pheromone gives two products: CH3(CH2)12COOH and CH3(CH2)7COOH. Suggest a structure for this sex attractant. Explainwhich part of the structure is uncertainarrow_forward1a)Give structures for the following compounds: a) phenyl isobutyl ether b) ethyl 4-phenylbenzoate c) 3-sec- butylpyridine d) 3-fluoro-3-hexen-2-onearrow_forwardFollowing is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker with high selectivity for airway smooth muscle receptors. Q. Propose reagents to bring about each step.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
